Inside the Ring Series 4: Peptides on a Mission — Delivering Drugs Where They Matter
Welcome to the Inside the Rings Series 4. Disclaimer: cyclic peptide is a light topic in this article as it focuses more on peptides.
Peptides are small but mighty. They’re rewriting the rules of drug delivery. From slipping past stubborn cell membranes to carrying powerful therapeutics exactly where they’re needed, these molecular couriers are turning scientific imagination into clinical reality.
The Journey of a Drug: Navigating a Hostile Landscape
The voyage of a therapeutic molecule through the human body is treacherous. Before it ever reaches its intended destination, it must face a myriad of biological barriers including enzymatic degradation, rapid clearance, and nonspecific distribution. The key question becomes: how do we ensure that a molecule survives this journey to successfully engage its target?
This is where drug delivery systems (DDSs) come in. More than just carriers, DDSs are engineered to shepherd therapeutics through this hostile terrain, protecting them from premature breakdown and misdirection. By minimizing systemic toxicity and reducing clearance, DDSs improve the odds of therapeutic success. DDSs also provide control by sustaining drug release over time, regulating spatial and temporal distribution, and fine-tuning dosing at the molecular level. The result is a higher effective concentration of the drug at the site of action to maximize efficacy while reducing collateral damage to the rest of the body. [1]
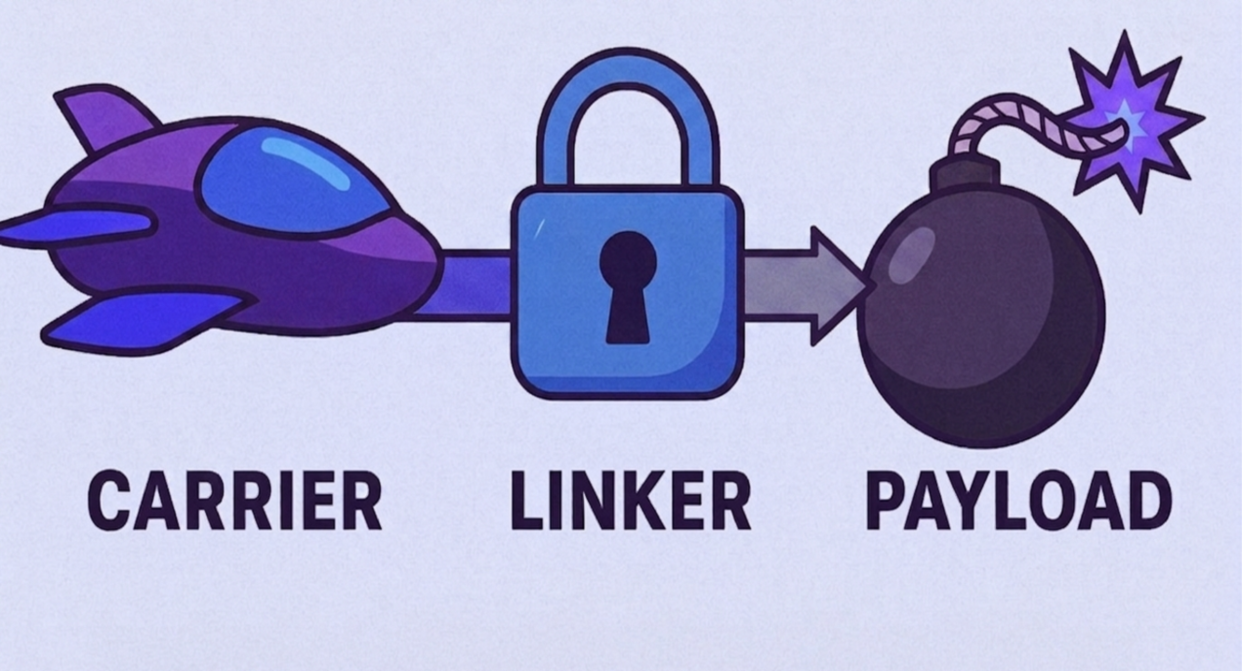
The Hero, the Weapon, and the Lock
One way to picture modern drug delivery is through a superhero movie:
- Carrier (the hero’s vehicle): A specialized vessel, like a spaceship or high-speed bike, designed to head straight for the villain’s hideout. This role is often played by antibodies, which can recognize and bind to disease markers.
- Payload (the weapon): A powerful but dangerous tool, too destructive to be released indiscriminately. This is often played by a highly potent chemotherapy drug that is safeguarded until it reaches the target.
- Linker (the lock): The secure connection between vehicle and weapon. It holds firm during transit but breaks apart once inside the villain’s base, releasing the drug at the exact right place and time.
Are Peptides the Right Vehicles?
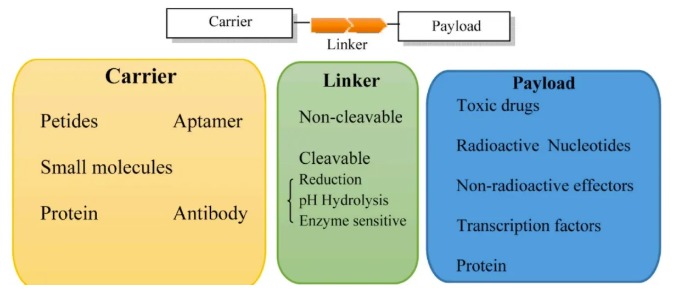
Peptides offer a compelling alternative to antibodies in this delivery framework. Defined by the FDA as polymers of ≤40 amino acids [2], they serve as a combination of favorable traits, like biocompatibility, tunability, stability, and ease of synthesis. Their side chains present a toolkit of reactive groups (carboxyl, hydroxyl, amino, thiol), enabling diverse chemical modifications for custom-designed delivery systems [3]. With high target affinity, low immunogenicity, and adjustable molecular size, peptides can be coupled to drugs via cleavable or stable linkers to form peptide–drug conjugates. These act as smart prodrugs engineered for precision release.
Peptide-based DDSs have already been harnessed to carry cytotoxic agents, nucleic acids, radionuclides, and more. They improve stability, reduce off-target toxicity, extend circulation times, and increase drug accumulation at disease sites. Meanwhile, advances in peptide display and synthesis platforms are driving innovation, with companies like Bicycle Therapeutics and PeptiDream Inc. at the forefront.
Different Peptides Act as Different Vehicles
Some peptides are precise navigators, while others are brute-force gate crashers, and still others act as clever shape-shifters that respond to their environment. Let’s take a closer look at the major classes.
Cell-Targeting Peptides (CTPs): The GPS System
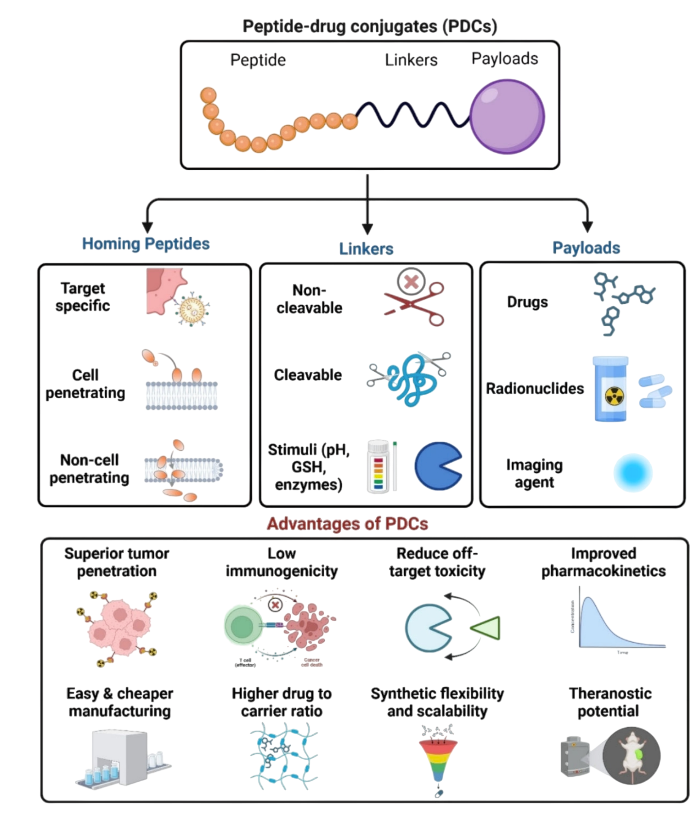
Drug delivery systems often exploit receptors that are over-expressed on diseased cells. Cell-targeting peptides (3–25 amino acids) function as homing devices, guiding drugs straight to their intended site. [4,5] Once bound to these receptors, they can internalize the therapeutic cargo and release it at the right spot.
This is the basis of peptide–drug conjugates (PDCs): a targeting peptide, a cleavable linker, and a cytotoxic payload. Here's the mechanism of action for narrowly defined PDCs: The peptide ligand binds to an over-expressed receptor on the surface of tumor cells, enabling receptor-mediated internalization and delivery of the cytotoxin into the malignant cell. Then, the linker undergoes cleavage, either triggered by intracellular or tumor-microenvironmental enzymes or by distinct chemical conditions, releasing the free drug to exert its full anti-tumor cytotoxic effect. [6] Compared with antibodies, peptides penetrate tissues more deeply, trigger fewer immune responses, and can be engineered with non-natural modifications for added stability and precision. [7-8]
Peptide Receptor Radionuclide Therapy (PRRT): The Radio Beacon
In PRRT, peptides are coupled to radionuclides and used to deliver radiation directly to tumors. Once bound, these peptide–receptor complexes either internalize or stay firmly attached, ensuring that radiation stays localized [9-10]. This approach has become a standard treatment for neuroendocrine tumors (NETs), with the FDA-approved drug Lutathera® paving the way. Although the approach was initiated over three decades ago, it has only been incorporated into major oncology guidelines in recent years after evidence of efficacy and safety in clinical trials. After the results of the phase III NETTER-1 trial in 2017, Luthatera® received approval for treatment of G1/G2 NET patients; the long-term results of the phase III NETTER-2 trial may offer a new first-line treatment option for advanced G2/G3 patients. [11]
Next-generation PRRT programs are in motion: PeptiDream Inc. and its partners (Novartis, Bristol Myers Squibb Eli Lilly are advancing a diverse pipeline of peptide radiopharmaceuticals, including promising candidates targeting CA9, CLDN18.2, and FAPα.
Peptide Ligands for Extrahepatic RNA Delivery
Antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs) silence gene expression by recruiting RNase H1 (ASOs) or Ago2 (siRNAs) to cleave complementary RNA sequences [12]. Several have been approved for clinical use [13,14], but thus far, their impact has been largely limited to the liver, eye, and CNS, organs where delivery is either naturally accessible or achieved by local administration.
In animal studies, systemically dosed ASOs distribute widely and show activity in many tissues, while siRNAs face poor bioavailability and restricted activity outside permissive compartments [15]. Targeted uptake can change this equation: GalNAc conjugation has revolutionized hepatocyte-directed delivery, providing clear advantages over unconjugated molecules [16]. Extending such precision targeting to extrahepatic tissues remains a challenge. This is where peptides stand out as promising ligands. Cyclized and engineered designs can reach nanomolar affinities, yet remain much smaller than antibodies — about 150-fold lower in molecular weight — allowing for lower doses, subcutaneous injection, and improved tissue penetration. They can also be modified with noncanonical amino acids to extend half-life, further boosting in vivo performance.
The industry is already moving in this direction. Alnylam Pharmaceuticals and PeptiDream Inc. have advanced peptide–siRNA conjugates, Ionis Pharmaceuticals, Inc. and Bicycle Therapeutics are developing peptide–ASO platforms for muscle, and Arrowhead Pharmaceuticals has taken a peptide–RNAi candidate (via its TRiM™ platform) into clinical trials. Together, these efforts highlight peptides as a frontier strategy for extrahepatic RNA delivery.
Cell-Penetrating Peptides (CPPs): The Gate Crashers
The cell membrane is nature’s security gate, great for protecting cells, but a formidable obstacle to drugs, proteins, or nucleic acids. That’s where cell-penetrating peptides (CPPs) are used. These short sequences (8–30 amino acids) can slip through membranes without tearing them apart. They act as molecular shuttles, carrying a wide range of cargo directly into the cell’s interior [17].
To get the cargo across, CPPs use two main routes (Figure 3):
- Direct translocation – Due to their positive charge, CPPs can briefly destabilize the membrane, creating temporary openings.
- Endocytosis – In this energy-driven pathway, the cell essentially engulfs the peptides into small vesicles. Depending on the conditions, this can happen through macropinocytosis, caveolin-mediated endocytosis (CvME), or clathrin-mediated endocytosis (CME) [20].
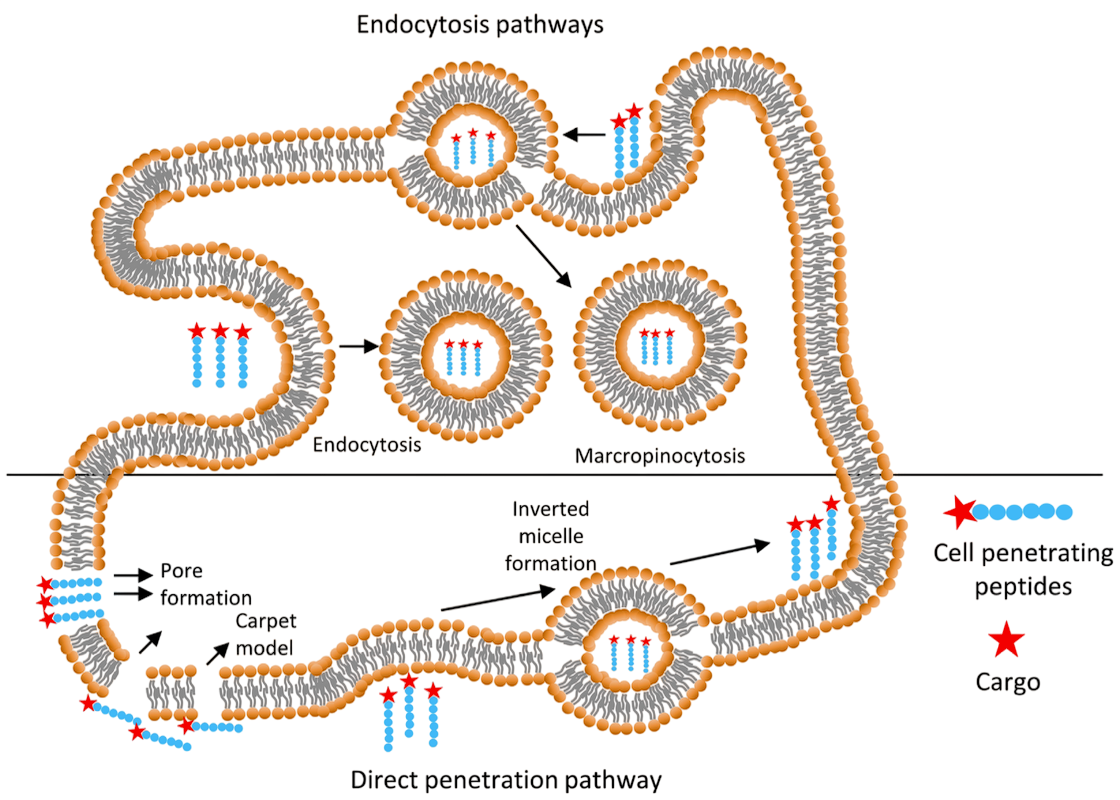
CPPs have turned what used to be a locked door into a two-way passage that opens new possibilities for drug delivery inside cells.
Stimuli-Responsive Peptides: The Shape-Shifters
The tumor microenvironment is acidic, enzyme-rich, and redox-active, a perfect opportunity for responsive peptides to adapt. These systems change charge, size, or conformation in response to local conditions and trigger smart drug release.
- Enzyme-responsive peptides (e.g., GFLG, YSA) release payloads only where tumor proteases are abundant.
- pH-responsive peptides exploit acidic conditions to enhance uptake and enable endosomal escape, a critical hurdle for RNA therapies. Platforms like Entrada’s EEV™ demonstrate how cyclic CPPs can dramatically improve intracellular delivery efficiency.
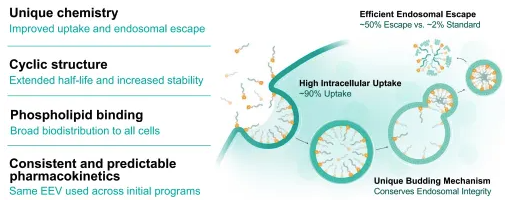
- Redox-responsive peptides sense elevated reactive oxygen species (ROS) or glutathione in tumors, cleaving or destabilizing themselves to release drugs selectively in diseased tissues.
Together, these adaptive systems transform peptides from passive carriers into active participants in drug delivery with precise release and improved therapeutic windows.
The Road Ahead: AI-Powered Peptide–Drug Conjugates
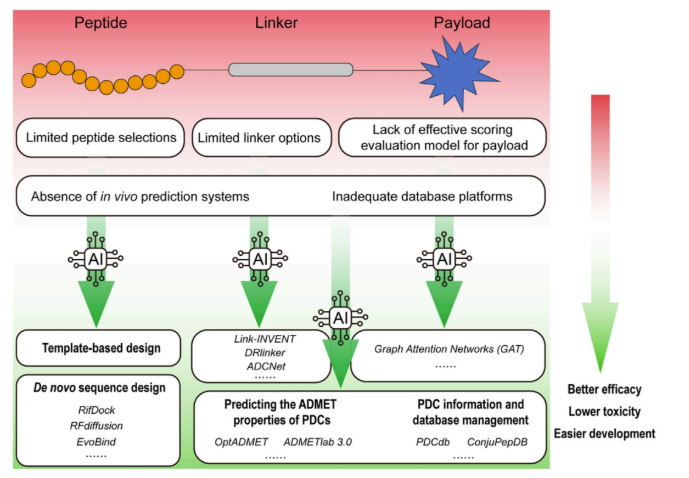
Despite decades of progress, peptide-based delivery still faces familiar roadblocks that can slow the drug journey: too few targeting ligands, limited linker options, weak payload scoring models, and poor in vivo prediction.
However, AI is changing the plot. For nearly a century, peptide discovery has relied on resource-heavy methods like SPPS, DELs, and phage or mRNA display. Now, with deep learning, tools like RFdiffusion have already produced cyclic tumor-targeting peptides with ~60% higher affinity than sequences from phage display, all while maintaining atomic-level precision (RMSD <1.5 Å). Recent models like RFPeptide are pushing this even further.
The momentum is undeniable. The 2024 Nobel Prize in Chemistry, awarded for AI-enabled protein design, signaled a clear shift: computational design has moved from a supporting role to a main character.
There's potential to streamline every layer of peptide–drug conjugate development:
- Ligand discovery - de novo generation of high-affinity targeting peptides
- Linker engineering - predictive modeling of stability and release
- Payload selection - AI-driven scoring for potency and safety
- ADME prediction - in silico testing of circulation, clearance, and biodistribution
AI not only makes peptide design faster, but also opens entirely new design spaces. What once took years of trial-and-error can now be achieved within weeks, bringing smarter, safer peptide–drug conjugates within reach.
Aknowledgments
Thank you to Long Pang (pangl@dp.tech), Dongdong Wang (wangdd@dp.tech), and Jin (Tina) Yu (yujin@dp.tech) for their valuable contributions to the content for this series.
References
[1] Agrawal M, Saraf S, Saraf S, Dubey SK, Puri A, Gupta U, Kesharwani P, Ravichandiran V, Kumar P, Naidu VGM, Murty US, Ajazuddin, Alexander A. Stimuli-responsive In situ gelling system for nose-to-brain drug delivery. J Control Release. 2020 Nov 10;327:235-265. doi: 10.1016/j.jconrel.2020.07.044. Epub 2020 Jul 31. PMID: 32739524.
[2] Varanko A, Saha S, Chilkoti A. Recent trends in protein and peptide-based biomaterials for advanced drug delivery. Adv Drug Deliv Rev. 2020;156:133-187. doi: 10.1016/j.addr.2020.08.008. Epub 2020 Aug 29. PMID: 32871201; PMCID: PMC7456198.
[3] Guo S, Wang J, Wang Q, Wang J, Qin S, Li W. Advances in peptide-based drug delivery systems. Heliyon. 2024 Feb 7;10(4):e26009. doi: 10.1016/j.heliyon.2024.e26009. PMID: 38404797; PMCID: PMC10884816.
[4] Majumdar S, Siahaan TJ. Peptide-mediated targeted drug delivery. Med Res Rev. 2012 May;32(3):637-58. doi: 10.1002/med.20225. Epub 2010 Sep 2. PMID: 20814957.
[5] Cooper BM, Iegre J, O' Donovan DH, Ölwegård Halvarsson M, Spring DR. Peptides as a platform for targeted therapeutics for cancer: peptide-drug conjugates (PDCs). Chem Soc Rev. 2021 Feb 15;50(3):1480-1494. doi: 10.1039/d0cs00556h. PMID: 33346298.
[6] Rizvi SFA, Zhang L, Zhang H, Fang Q. Peptide-Drug Conjugates: Design, Chemistry, and Drug Delivery System as a Novel Cancer Theranostic. ACS Pharmacol Transl Sci. 2024 Jan 24;7(2):309-334. doi: 10.1021/acsptsci.3c00269. PMID: 38357281; PMCID: PMC10863443.
[7] David A. Peptide ligand-modified nanomedicines for targeting cells at the tumor microenvironment. Adv Drug Deliv Rev. 2017 Sep 15;119:120-142. doi: 10.1016/j.addr.2017.05.006. Epub 2017 May 12. PMID: 28506743.
[8] Vadevoo SMP, Gurung S, Lee HS, Gunassekaran GR, Lee SM, Yoon JW, Lee YK, Lee B. Peptides as multifunctional players in cancer therapy. Exp Mol Med. 2023 Jun;55(6):1099-1109. doi: 10.1038/s12276-023-01016-x. Epub 2023 Jun 1. PMID: 37258584; PMCID: PMC10318096.
[9] Bodei L, Pepe G, Paganelli G. Peptide receptor radionuclide therapy (PRRT) of neuroendocrine tumors with somatostatin analogues. Eur Rev Med Pharmacol Sci. 2010 Apr;14(4):347-51. PMID: 20496546.
[10] Murray D, McEwan AJ. Radiobiology of systemic radiation therapy. Cancer Biother Radiopharm. 2007 Feb;22(1):1-23. doi: 10.1089/cbr.2006.531. PMID: 17627411.
[11] Santo G, Di Santo G, Virgolini I. Peptide Receptor Radionuclide Therapy of Neuroendocrine Tumors: Agonist, Antagonist and Alternatives. Semin Nucl Med. 2024 Jul;54(4):557-569. Doi: 10.1053/j.semnuclmed.2024.02.002. Epub 2024 Mar 15. PMID: 38490913.
[12] Setten RL, Rossi JJ, Han SP. The current state and future directions of RNAi-based therapeutics. Nat Rev Drug Discov. 2019 Jun;18(6):421-446. doi: 10.1038/s41573-019-0017-4. Erratum in: Nat Rev Drug Discov. 2020 Apr;19(4):291. doi: 10.1038/s41573-019-0023-6. Erratum in: Nat Rev Drug Discov. 2020 Apr;19(4):290. doi: 10.1038/s41573-019-0027-2. PMID: 30846871.
[13] Rinaldi C, Wood MJA. Antisense oligonucleotides: the next frontier for treatment of neurological disorders. Nat Rev Neurol. 2018 Jan;14(1):9-21. doi: 10.1038/nrneurol.2017.148. Epub 2017 Dec 1. PMID: 29192260.
[14] Egli M, Manoharan M. Chemistry, structure and function of approved oligonucleotide therapeutics. Nucleic Acids Res. 2023 Apr 11;51(6):2529-2573. doi: 10.1093/nar/gkad067. PMID: 36881759; PMCID: PMC10085713.
[15] Hung G, Xiao X, Peralta R, Bhattacharjee G, Murray S, Norris D, Guo S, Monia BP. Characterization of target mRNA reduction through in situ RNA hybridization in multiple organ systems following systemic antisense treatment in animals. Nucleic Acid Ther. 2013 Dec;23(6):369-78. doi: 10.1089/nat.2013.0443. Epub 2013 Oct 26. Erratum in: Nucleic Acid Ther. 2014 Apr;24(2):178. PMID: 24161045.
[16] Prakash TP, Graham MJ, Yu J, Carty R, Low A, Chappell A, Schmidt K, Zhao C, Aghajan M, Murray HF, Riney S, Booten SL, Murray SF, Gaus H, Crosby J, Lima WF, Guo S, Monia BP, Swayze EE, Seth PP. Targeted delivery of antisense oligonucleotides to hepatocytes using triantennary N-acetyl galactosamine improves potency 10-fold in mice. Nucleic Acids Res. 2014 Jul;42(13):8796-807. doi: 10.1093/nar/gku531. Epub 2014 Jul 3. PMID: 24992960; PMCID: PMC4117763.
[17] Kalmouni M, Al-Hosani S, Magzoub M. Cancer targeting peptides. Cell Mol Life Sci. 2019 Jun;76(11):2171-2183. doi: 10.1007/s00018-019-03061-0. Epub 2019 Mar 15. PMID: 30877335; PMCID: PMC11105397.
[18] Ouyang J, Sheng Y, Wang W. Recent Advances of Studies on Cell-Penetrating Peptides Based on Molecular Dynamics Simulations. Cells. 2022 Dec 12;11(24):4016. doi: 10.3390/cells11244016. PMID: 36552778; PMCID: PMC9776715.
[19] de Jong H, Bonger KM, Löwik DWPM. Activatable cell-penetrating peptides: 15 years of research. RSC Chem Biol. 2020 Aug 26;1(4):192-203. doi: 10.1039/d0cb00114g. PMID: 34458758; PMCID: PMC8341016.
[20] Reid LM, Verma CS, Essex JW. The role of molecular simulations in understanding the mechanisms of cell-penetrating peptides. Drug Discov Today. 2019 Sep;24(9):1821-1835. doi: 10.1016/j.drudis.2019.06.013. Epub 2019 Jun 20. PMID: 31229665.
[21] Zhang DE, He T, Shi T, Huang K, Peng A. Trends in the research and development of peptide drug conjugates: artificial intelligence aided design. Front Pharmacol. 2025 Feb 27;16:1553853. doi: 10.3389/fphar.2025.1553853. PMID: 40083376; PMCID: PMC11903715.