Inside the Ring Series 3: Prioritizing Immunology Targets for Cyclic Peptides

Over more than a century of development, peptide therapeutics have steadily gaining recognition for their significant clinical value. Industry momentum spurred market expansion and positioned peptides, especially cyclic peptides, as a core modality in modern drug discovery. Among the six peptide drugs approved in 2023, half were cyclic (Rezafungin, Motixafortide, Zilucoplan), a striking testament to their importance (check out Inside the Ring Series 1.5).
The sector is also attracting significant investment: in January 2024, Merck entered a $220 million collaboration with Unnatural Products to advance cyclic peptide therapeutics. Just a month later, Orbis Medicines, incubated by Novo Nordisk, secured 90 million Euro in Series A funding. These developments signal that cyclic peptides are evolving into central pillars of the next industrial upgrade in peptides.
Cyclic Peptides Lead Therapeutic Frontiers
Cyclic peptides are defined by their closed ring structures that provide pharmacological advantages like enhanced stability, improved selectivity, and higher affinity binding to protein–protein interfaces. But how do these features translate into clinical impact?
Recent clinical programs highlight their versatility:
- Metabolic disease: Merck’s MK-0616, a first-in-class oral PCSK9 inhibitor, has met Phase 3 endpoints in CORALreef HeFH and AddOn studies, addressing a longstanding need for convenient therapies in hypercholesterolemia.[1]
- Oncology: Roche’s LUNA18, an oral KRAS G12D inhibitor, leverages cyclic peptide design to tackle one of the most elusive oncogenic drivers. Structural optimization has improved oral bioavailability to 21–47% in preclinical models.[2]
- Immunology: Johnson & Johnson’s JNJ-77242113, the first oral cyclic peptide inhibitor of the IL-23 pathway, has shown positive signals in Phase 3, validating the modality’s potential in immune-mediated disease.[3]
Immunology Primed for Innovation
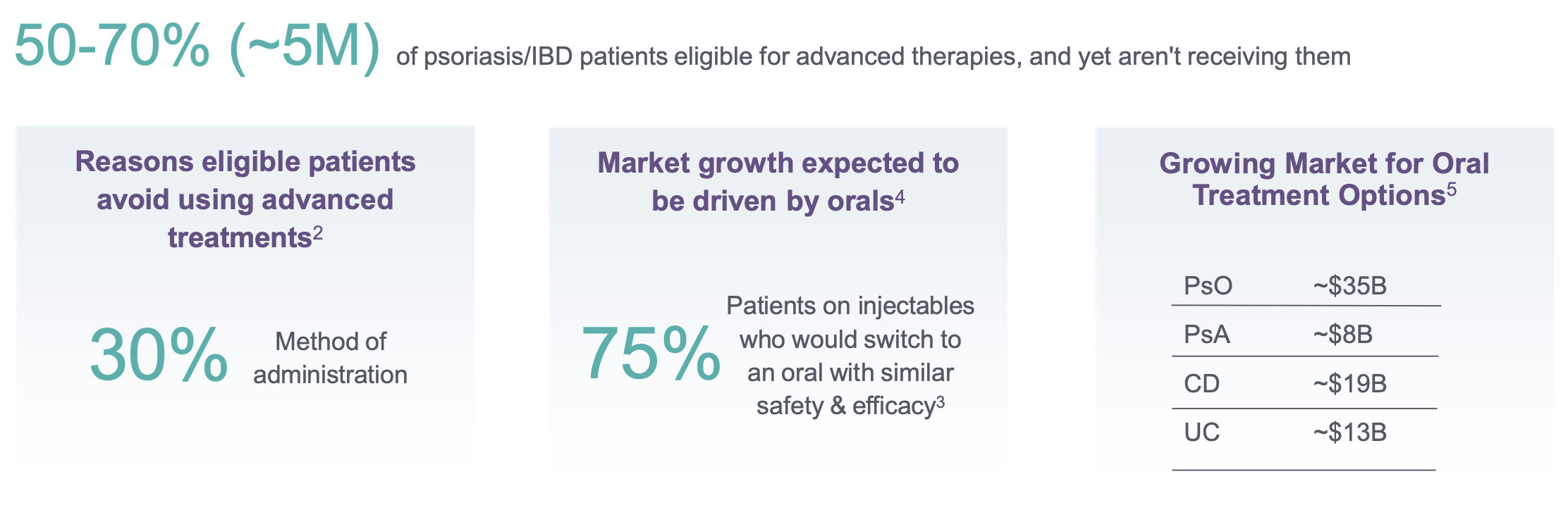
Among the therapeutic areas, immunology is arguably the most fertile ground for cyclic peptide innovation. Unmet need is high: oral therapy adoption in psoriasis and inflammatory bowel disease (IBD) are still limited, with 50–70% of eligible patients undertreated despite available biologics. Surveys indicate that, when efficacy and safety are comparable, up to 75% of antibody-treated patients would prefer an oral alternative.
The biology reinforces the opportunity. Immune-related targets frequently involve flat, extended PPIs that small molecules struggle to engage. Biologics provide sufficient affinity but are restricted by parenteral delivery. Cyclic peptides fill this gap because they're structurally capable of mimicking antibody binding yet compact enough to be optimized for oral formulations. In immunology, the right targets could redefine standard of care.
A Framework for Prioritizing Targets
To guide investment and development, we propose five key criteria to identify immunology targets well-suited for cyclic peptide intervention:
1. The Oral Imperative: Meeting a Pressing Clinical Need
Patient adherence reveals a critical gap in current therapeutics. According to Johnson & Johnson's commercial research, 50–70% of patients with psoriasis and inflammatory bowel disease remain under-treated due to injection burden and risk-benefit concerns. When efficacy is comparable, 75% of current antibody-treated patients would switch to oral alternatives, reflecting a clear preference for integrated, daily-life-compatible treatments. By 2030, oral treatments targeting the IL-23/IL23R axis across psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis are projected to reach $75 billion in market share. This emphasizes a fundamental shift toward patient-centric therapeutic delivery that matches biologic efficacy while dramatically improving treatment experience.
2. Achieving Excellence: Focus on Validated Therapeutics
This rule centers on two strategically selected categories: monoclonal antibodies and agents with robust Phase 2/3 validation against well-characterized targets. Consider TL1A (Tumor necrosis factor-like ligand 1A), a member of the TNF superfamily that exemplifies this approach: when TL1A engages its receptor DR3, it triggers a TRADD-dependent signaling cascade that orchestrates the secretion of pro-inflammatory cytokines and chemokines, ultimately driving inflammation and fibrosis. While all clinical-stage TL1A programs to date have used antibody modalities, with the most advanced reaching Phase 3 in inflammatory bowel disease, the target itself represents validated biology with proven therapeutic potential.
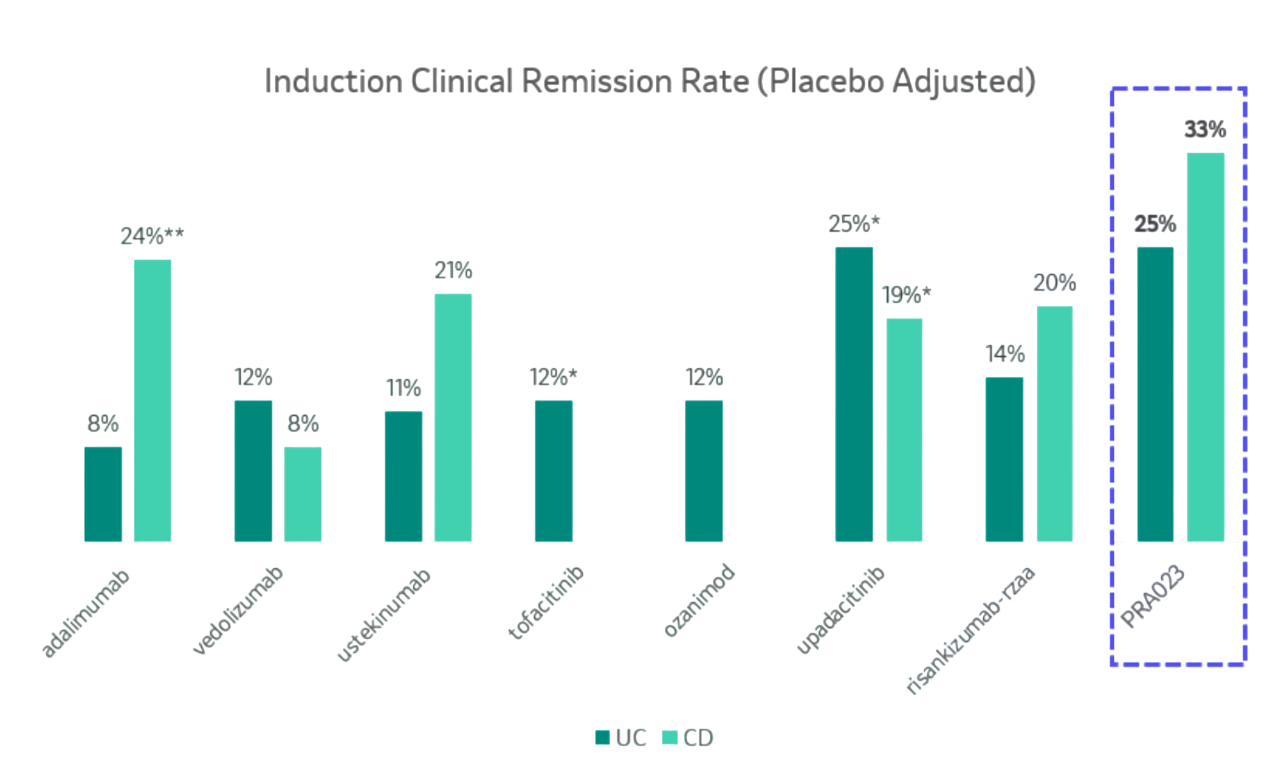
The clinical data speaks volumes: anti-TL1A monoclonal antibodies have delivered outstanding Phase 2 results (Figure 2), and in indirect comparisons, their efficacy profiles appear highly competitive within their respective indications. This combination of validated mechanism, advanced clinical stage, and demonstrated efficacy creates precisely the kind of de-risked opportunity.
3. Opening Window: Low Small Molecule Competition
The current therapeutic landscape for these validated targets reveal a striking pattern: antibodies dominate, while oral alternatives are relatively absent. Small molecule and peptide programs either don't exist at the clinical stage or are significantly less efficacious than their antibody counterparts. For example, despite robust antibody validation, there are no TL1A oral drugs currently in clinical development. PharmaCube pipeline data shows only three preclinical small molecule or peptide programs targeting TL1A, and similar patterns across targets like TSLP, where antibody programs overwhelmingly dominate development pipelines.
This creates what we call "antibody-to-oralizable" targets — validated biology with minimal oral competition and limited competitive pressure. These targets often lack classical small-molecule binding pockets, making them suited for constrained peptide formats like cyclic peptides, and creating a window for strategic investment in oral therapeutics targeting validated, antibody-proven mechanisms.
4. Strategic Investment: Value Through Validation and Scale
Precedent products across our target portfolio validate the oral therapeutics opportunity:
- Scale and Growth Trajectory: These indications address substantial, expanding patient populations with clear unmet need. IBD alone affects 6–8 million individuals worldwide[4].
- Chronic Care Economics: The lifelong nature of these conditions creates exceptional lifetime treatment value. For instance, psoriasis patients require continuous management, making patient-friendly oral administration not just preferable, but also economically essential for sustained adherence and treatment success.
- Proven Commercial Validation: Existing therapeutics provide tangible proof of market potential. For example, Tezepelumab, the first-in-class TSLP-targeting antibody for severe asthma, has achieved blockbuster status with rapid revenue growth [5].
- Expandable Biology: Targets with biology relevant to multiple disease contexts amplify the addressable market. Beyond IBD, TL1A biology extends to rheumatoid arthritis, atopic dermatitis, pulmonary fibrosis, and intestinal fibrosis, each an additional revenue stream from a single therapeutic investment.
This systematic approach to target selection mirrors the strategic framework underlying our cyclic peptide platform: not just developing molecules, but architecting portfolio value through validated, scalable opportunities.
5. Structural Intelligence for Rational Design
Clear target structure dramatically lowers the barrier to successful drug design. When existing antibody therapeutics provide additional epitope mapping, the advantage becomes even more pronounced. Without experimental structural data, computational predictions can guide development—though this typically extends timelines and reduces success probability.
Case Study: TL1A as a Model Target
TL1A (TNFSF15) demonstrates how this framework can be applied.
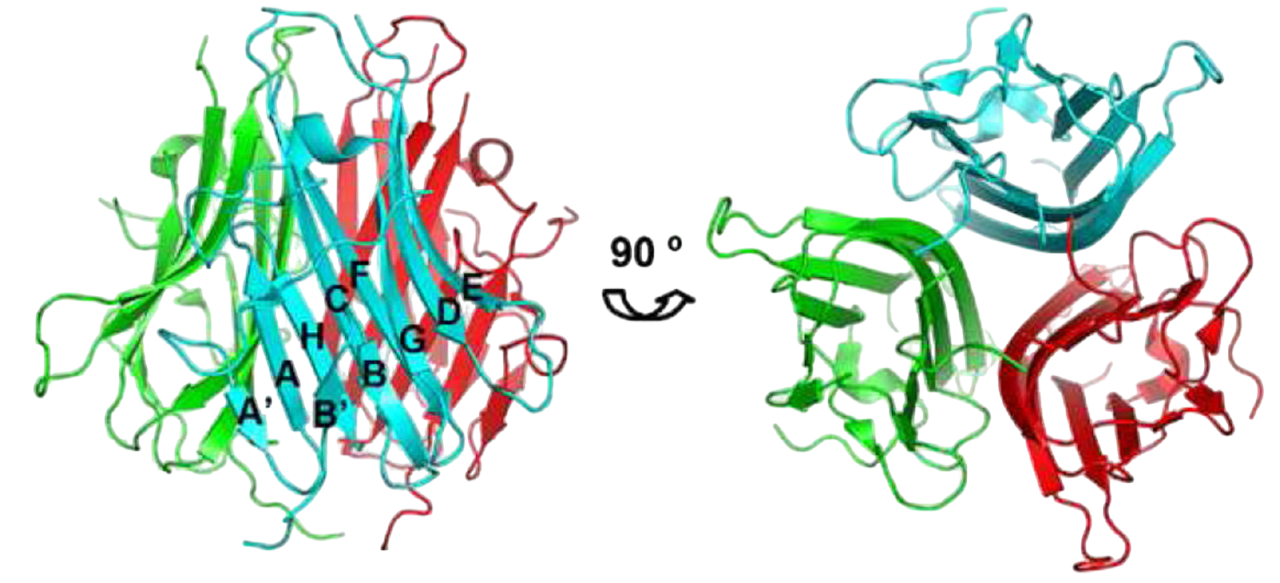
TL1A is an ideal structural target. As a TNF superfamily member (TNFSF15), it's expressed across immune cells — monocytes, macrophages, dendritic cells, and T cells — plus non-immune types like synovial fibroblasts and endothelial cells. The protein exists in both membrane-bound (mTL1A) and soluble (sTL1A) forms, with its C-terminal region containing the canonical TNF homology domain that assembles into a well-characterized trimeric structure. TL1A's functional receptor DR3 is mainly found on activated lymphocytes. The TL1A-DR3 interaction forms a defined 3:3 trimer-to-trimer complex, providing clear structural guidance for drug design.
Pathway Complexity Unveils Therapeutic Opportunity
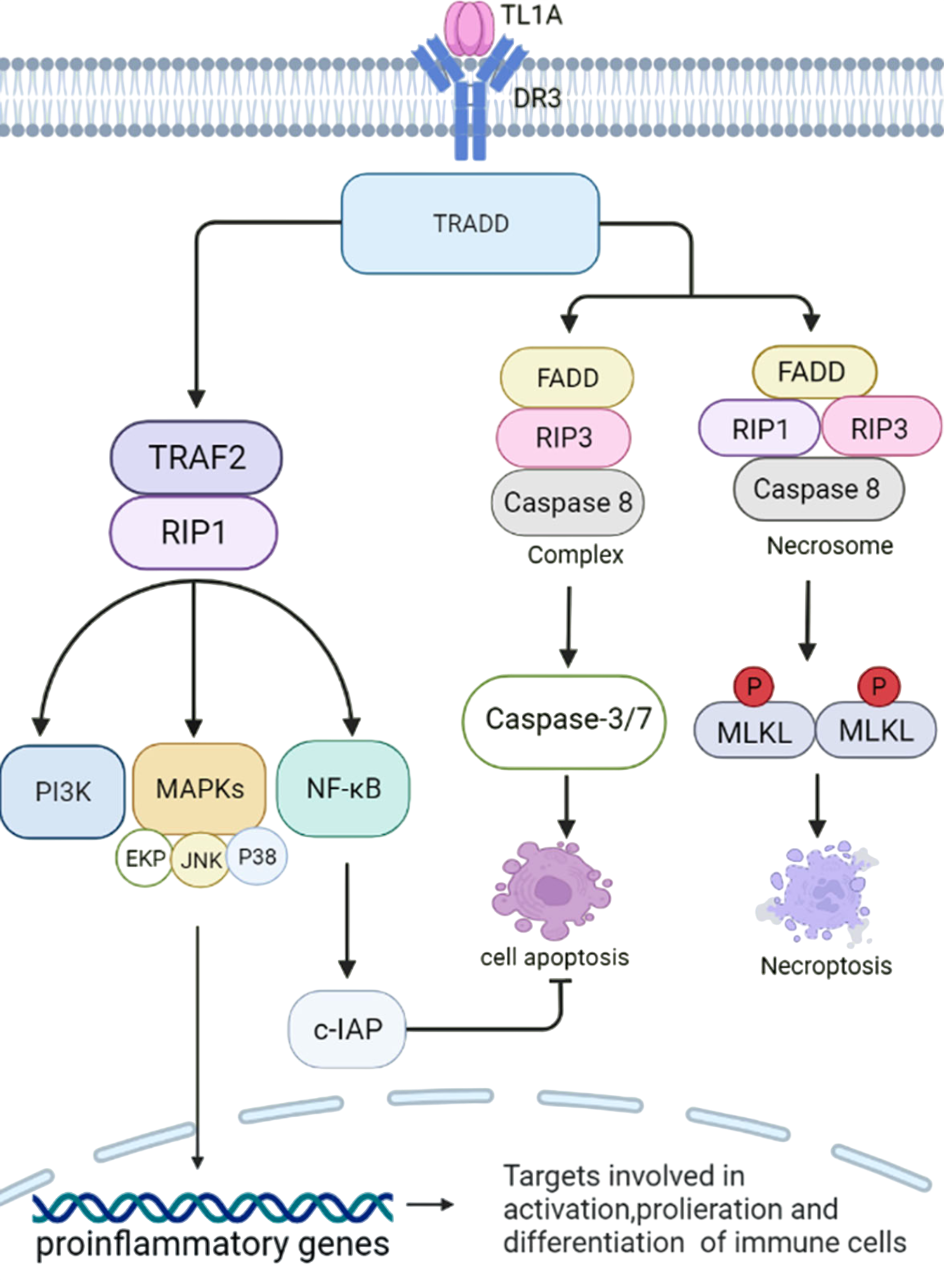
TL1A binds to DR3 to trigger multiple signaling cascades with distinct outcomes. The primary pathway recruits TRADD, scaffolding TRAF2, RIP1, and cIAP1/2 to activate NF-κB, MAPK, and PI3K signaling, driving pro-inflammatory gene expression and cell proliferation. Alternative pathways can lead to caspase-dependent apoptosis or necroptotic cell death through necrosome formation. Moreover, DcR3 provides natural regulation by sequestering TL1A away from DR3 and reducing inflammatory signaling.
Elevated TL1A/DR3 expression is consistently observed in inflamed IBD patient tissue. Transgenic mice overexpressing TL1A develop ileal inflammation, while TL1A/DR3 knockout mice show significant protection from chronic colitis. Mechanistically, TL1A amplifies inflammatory T helper subsets (Th1/Th17), driving release of key cytokines including IFN-γ, TNF-α, IL-6, and IL-17. [8,9]
Summarizing the Market Potential:
- Inflammatory bowel disease (IBD), including ulcerative colitis, Crohn's disease, and indeterminate colitis, represents chronic intestinal inflammation driven by dysregulated immune responses. While the precise etiology remains complex, involving genetic, environmental, and immune factors, TL1A emerges as a central player in IBD pathogenesis.
- Expanding Market Opportunity: The treatment market was valued at $22.6 billion in 2023 and is projected to reach $38.2 billion by 2032 (6.1% CAGR).[10]
- Clear Unmet Need: Despite market size, therapeutic gaps persist. Approximately 40% of patients lose response to current biologics over time, 80% experience chronic relapse, and 20-30% require surgical resection. [12-13]
- The Oral Advantage: Patient preference studies consistently favor oral formulations, particularly among those with mild symptoms, positioning oral agents for superior adoption and adherence compared to injectable alternatives.[14]
Overall, TL1A represents an “antibody-to-oralizable” target: validated by antibodies, biologically central, but lacking oral alternatives, and is a perfect proving ground for cyclic peptides. In the next article, we'll delve into the sophisticated delivery systems that transform these molecules into clinically viable drugs.
Acknowledgments
Thank you to 聂方园 (niefangyuan@dp.tech), Dongdong Wang (wangdd@dp.tech), and Jin(Tina) Yu (yujin@dp.tech) for their valuable contributions in preparing the content for this series.
References
[2] Tanada. M., Tamiya M., Matsuo A., et al., Development of Orally Bioavailable Peptides Targeting an Intracellular Protein: From a Hit to a Clinical KRAS Inhibitor. J. Am. Chem. Soc. 2023 Aug 2;145(30):16610-16620.
[3] Fourier, A.M., et al. JNJ-77242113, a highly potent, selective peptide targeting the IL-23 receptor, provides robust IL-23 pathway inhibition upon oral dosing in rats and humans. Sci. Rep. 2024 Jul 30;14(1):17515.
[4] Mak W.Y., Zhao M., Ng S.C., et al. The epidemiology of inflammatory bowel disease: East meets westJ. Gastroenterol. Hepatol. 2020 Mar;35(3):380-389.
[5] https://www.phirda.com/artilce_36665.html?module=trackingCodeGenerator.
[6]Zhan C., Yan Q., Patskovsky Y., et al. Biochemical and Structural Characterization of the Human TL1A Ectodomain.Biochemistry. 2009 Aug 18; 48(32): 7636–7645.
[7] Xu W.D., Li R., huang A.F., et al., Role of TL1A in Inflammatory Autoimmune Diseases: A Comprehensive Review. Front. Immunol. 2022;14:13:891328.
[8] Richard A.C., Ferdinand J.R., Meylan F., et al.,The TNF-family cytokineTL1A: from lymphocyte co-stimulator to disease co-conspirator. J. Leukoc. Biol. 2015;98:333–345.554 G.
[9] Kokkotis G., Bamias G. TL1A as a therapeutic target in inflammatory bowel disease. Expert Rev. of Clin. Immunol. 2022;18:551–555.
[10] https://www.gminsights.com/industry-analysis/inflammatory-bowel-disease-treatment-market.
[11] Wang R., Li A., Liu S., et al. Global, regional and national burden of inffammatory bowel disease in 204 countries and territories from 1990 to 2019: a systematic analysis based on the Global Burden of Disease Study 2019.BMJ Open 2023;13:e065186.
[12] Sing S., George J., Boland B., et al. Primary non-response to tumor necrosis factor antagonists is associated with inferior response to second-line biologics in patients with Inflammatory bowel diseases: A systematic review and meta-analysis. J. Crohns Clitis 2018; 12:635–643.
[13] Nakase H. Treatment of inflammatory bowel disease from the immunological perspective, Immunol. Med. 2020 43:2, 79-86.
[14] Yasmin F., Najeeb H., Shaikh S., et al., Novel drug delivery systems for inflammatory bowel disease.World J. Gastroenterol. 2022 May 14; 28(18): 1922-1933.
[15] Kobayashi T., Mizuno N., Sato N., et al., Patient preferences for advanced therapies in ulcerative colitis using conjoint analysis.Intest. Res. 2024 October 14.