Inside the Ring Series 2: Cracking the Code of Permeability

There's no doubt that cyclic peptides are remarkable molecules capable of targeting the “undruggable” space where small molecules and biologics often fall short. However, there’s a catch: to reach their targets, cyclic peptides must first cross the cellular membrane. Without enough permeability, it wouldn't matter how promising the scaffold is.
In small-molecule discovery, Lipinski’s “Rule of Five” gave chemists a compass for designing orally available drugs. We set out to answer the question: Could cyclic peptides benefit from an equivalent set of guiding principles? By curating and analyzing multiple large-scale cyclic peptide datasets — nearly 30,000 molecules in total — we began to map how key physicochemical properties relate to permeability.
Mapping the Molecular Landscape: Flagish Cyclic Peptide Datasets
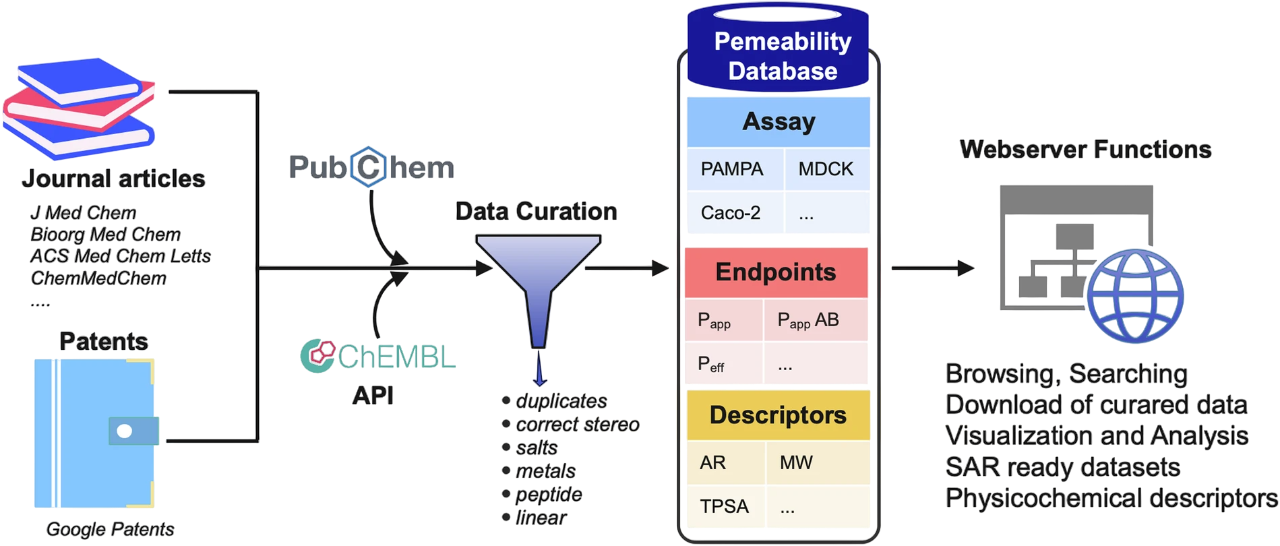
Understanding how cyclic peptides behave in biological systems requires robust, experimentally grounded data. Here, we introduce a few cyclic peptide datasets that will provide a molecular atlas for the field, including experimentally validated membrane permeability data covering over 9,000 cyclic peptides. These measurements span diverse assay types, like synthetic membrane systems (PAMPA) to cell-based models (Caco-2, MDCK, and RRCK), each offering unique insights on how these molecular rings navigate biological barriers.
CycPeptMPDB: The Curated Visualizer
When it comes to navigating the complex world of cyclic peptides, researchers need high-quality, relevant, and accessible literature. Enter CycPeptMPDB (http://cycpeptmpdb.com/), a meticulously curated database developed by Professor Yutaka Akiyama's team at the Tokyo Institute of Technology . This digital repository aggregates around 8,000 cyclic peptides from published articles and patents, organized around two core modules: peptide-level data (experimental permeability, sequences, structures, and physicochemical properties) and monomer fragment data (individual building blocks with their associated properties).
Rather than drowning users in raw permeability data from four different assay methods (PAMPA, Caco-2, MDCK, and RRCK), CycPeptMPDB takes a pragmatic approach in which experimental permeability is reported as LogPexp, with a detection floor at −10 (corresponding to Pexp = 1.0×10⁻¹⁰ cm/s).
Here's where clever design meets practical utility: peptides are color-coded using a LogPexp cutoff of −6.0. Those above this threshold are marked yellow for good permeability, and those below are marked green for low permeability (see figure 1 "Statistics"). It's a simple visual system that instantly communicates what matters most to drug discovery professionals.
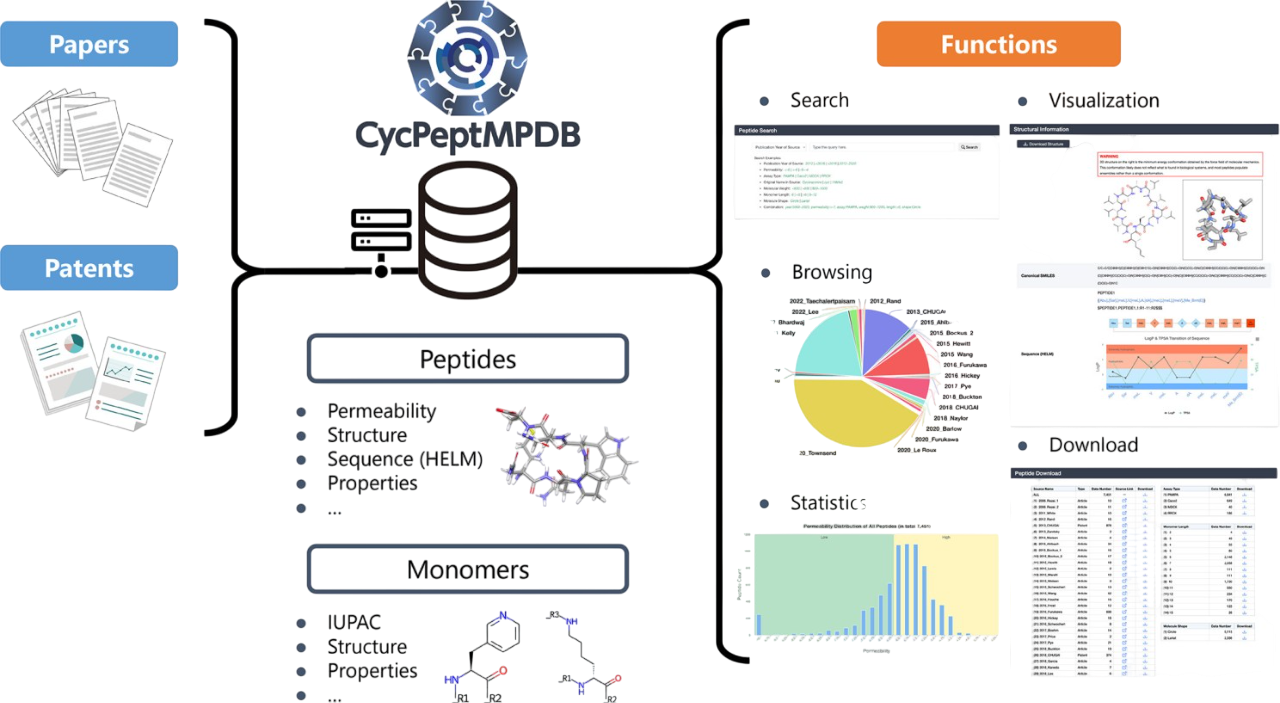
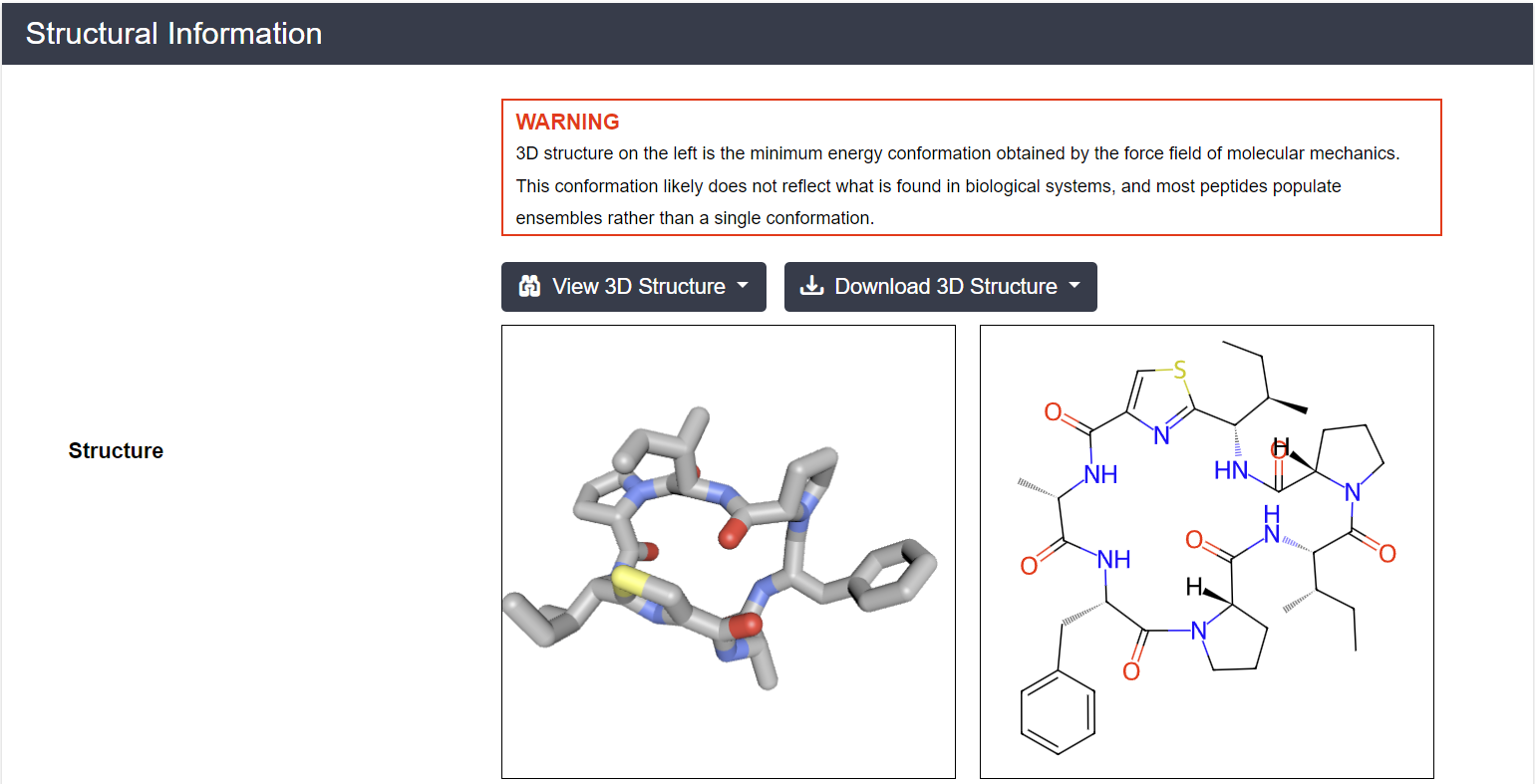
Under the hood, CycPeptMPDB uses sophisticated computational workflows to bring these molecules to life. Starting with RDKit-generated conformers (5,000 per peptide), the database filters for the most stable structures by eliminating redundancy using RMSD analysis and energy minimization. Under a quantum-mechanical protocol developed by Dr. Richard A. Lewis at Novartis, each peptide exists in two molecular worlds of water and chloroform, reflecting the dual nature of biological barriers these molecules must navigate. These 3D structures are interactive, visualizable, and downloadable resources that transform static sequences into dynamic molecular reality as shown in the figure 2.
NPMMPD: The Non-Peptidic Macrocycle Specialist
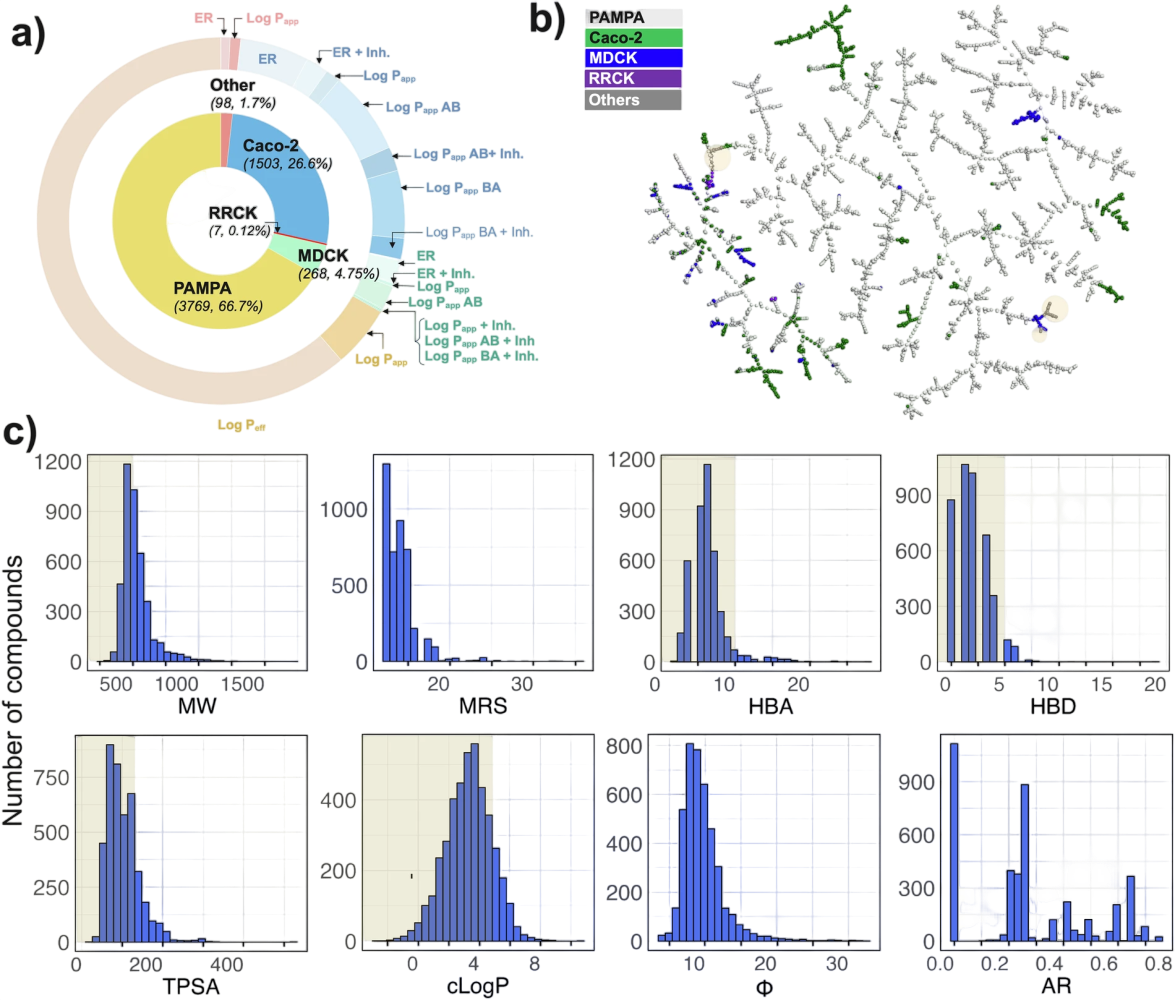
Cyclic peptides are just one chapter in the macrocycle story. NPMMPD (https://swemacrocycledb.com/), developed by Jan Kihlberg's research group at Uppsala University, Sweden, broadens the narrative to encompass the entire universe of non-peptidic macrocycles. This database represents a massive curation effort, consolidating data from 103 scientific publications and 9 patents from 2006 to 2023, plus entries from the ChEMBL repository to offer a collection of 5,638 permeability measurements across 4,612 macrocyclic molecules.
The database reveals patterns on how researchers study macrocycle permeability. PAMPA leads the charge with 67% of all measurements (3,767 entries), though notably, 91% of these PAMPA data points trace back to a single comprehensive publication. Caco-2 cell assays contribute another 26% (1,502 entries), capturing both directional permeability and efflux ratios to assess active transport mechanisms. MDCK measurements round out the major contributors with 264 entries.
Most importantly, this collection challenges conventional boundaries. Half of the compounds exceed 500 Da molecular weight. The majority still respect traditional Lipinski-like limits for polarity and lipophilicity. And 9% venture into true "beyond Rule of Five" territory, a potentially valuable chemical space for tackling challenging or "undruggable" targets.
The team's comprehensive molecular analysis, visualized through TMAP (Tree-MAP), reveals a structurally diverse landscape that range from drug-like molecules to more exotic macrocyclic architectures. Figure 4 breaks down permeability data by assay type and endpoints, TMAP visualization maps the structural landscape of 4,216 non-peptide macrocycles, and 2D descriptors capture molecular property distributions across size, polarity, lipophilicity, flexibility, and amide ratios, while Rule of Five and Veber rule boundaries provide context for just how far these molecules venture beyond conventional drug-like space.
Looking Beyond the Core Databases
Literature Enrichments
Several landmark studies have contributed to the field with substantial experimental datasets. Chugai Pharmaceutical Co., Ltd.'s 2023 JACS breakthrough on oral drug design for "undruggable" targets compiled 727 cyclic peptides, with 584 featuring experimental permeability data. A pivotal 2016 Nature Chemical Biology study explored structure-permeability relationships across 214 cyclic compounds, while a 2022 Cell paper on de novo macrocycle design contributed measurements for 196 cyclic peptides spanning various ring sizes.
Macrocycle-DB: The Panoramic Lens
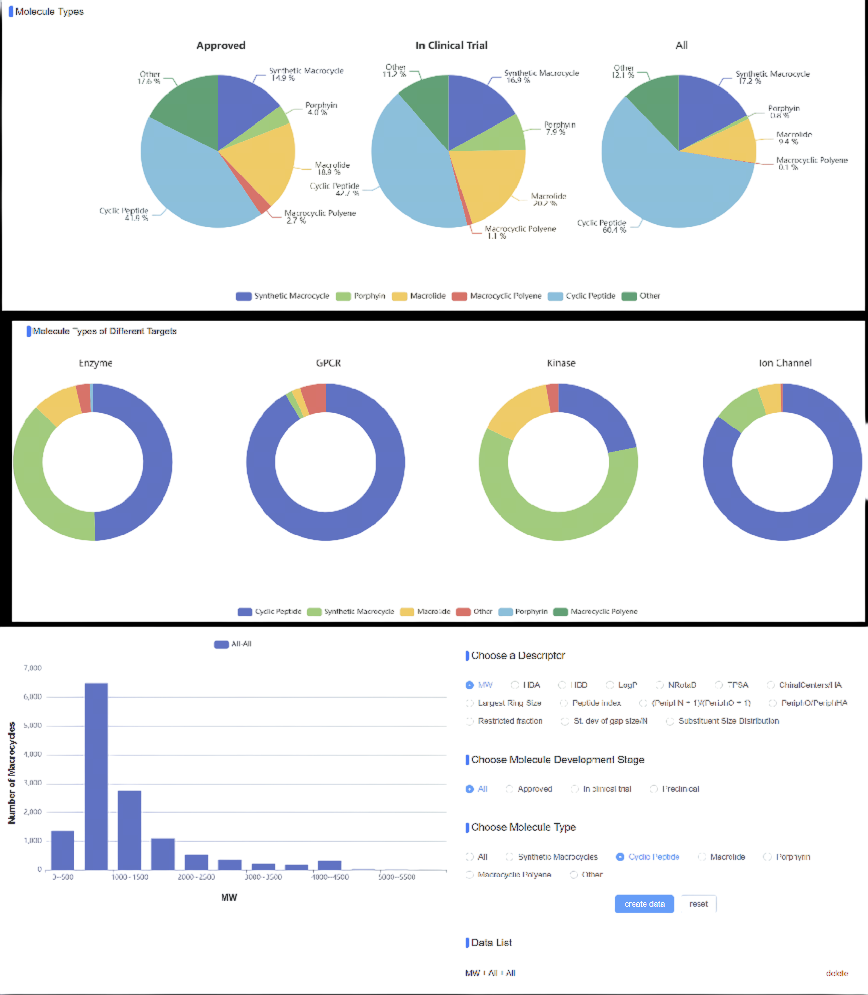
Macrocycle-DB (https://macro-db.dpbio.tech/) embodies a broad perspective, aggregating over 18,000 synthetic macrocycles from major repositories including PubChem, ChEMBL, and clinical databases. While encompassing diverse scaffolds including macrolides, porphyrins, and macropolyenes, cyclic peptides dominates not only as 61% of the collection, but also as 42% of marketed and clinical-stage molecules, virtually monopolizing GPCR and ion channel therapeutics.
Beyond cataloging, Macrocycle-DB computes 48 comprehensive molecular descriptors:
- Overall properties of macrocyclic compounds (1–14)
- Atom type distributions within the largest ring (15–21)
- Distributions of distinct bond types in the largest ring (22–25)
- Features of peripheral substituents (26–30)
- Substituent atom type composition (31–35)
- Substituent size and spatial distribution (36–40)
- Substituent spacing and distribution metrics (41–48)
CyclicPepedia: The Expert in Biological Diversity
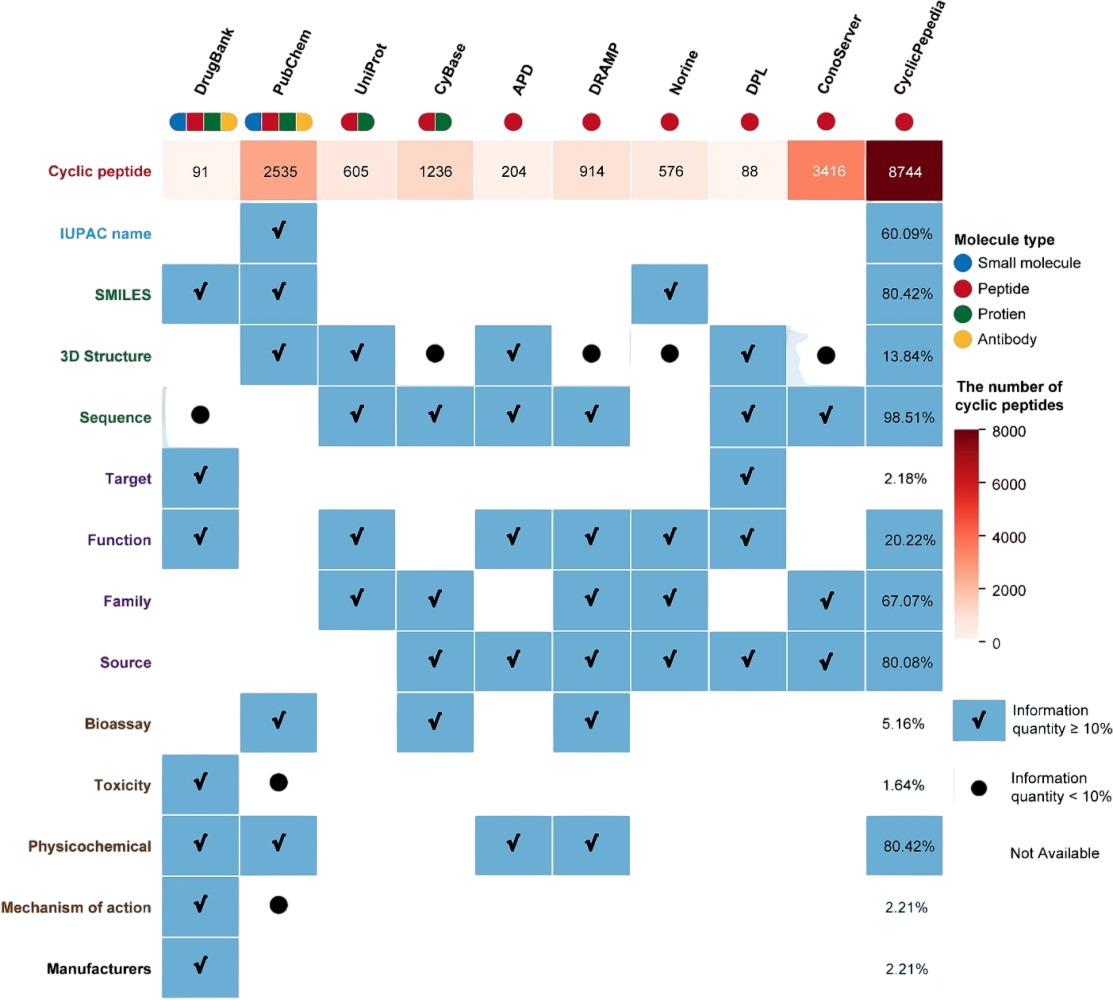
CyclicPepedia (https://www.biosino.org/iMAC/cyclicpepedia/) practices a different philosophy: embracing the natural diversity of cyclic peptides. Following its 2024 update, this resource catalogs 8,751 natural and synthetic cyclic peptides from 821 biological systems, organized into 180 families across 59 targets. Though lacking in experimental permeability data, CyclicPepedia excels in biological context. Drawing from 9 source databases, it provides sequence information for 8,614 entries and structural data for 7,032, with 1,210 featuring 3D structures from PubChem, PDB, and AlphaFold2.
Cracking the Permeability Code
Understanding the relationship between molecular structure and membrane permeability remains one of the most pressing challenges in cyclic peptide drug discovery. Using the CycPeptMPDB dataset, we uncovered several surprising patterns.
Size Matters
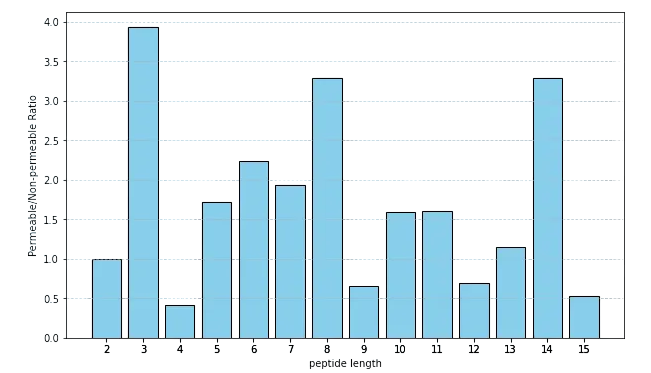
The CycPeptMPDB collection spans peptides from 2 to 15 residues, with hexapeptides, heptapeptides, and decapeptides leading the pack (2,099, 2,070, and 1,728 molecules, respectively). But things got interesting when we calculated the ratio of high-permeability to low-permeability molecules for each peptide length. Tripeptides emerged as permeability champions with a ratio of almost 4, likely benefiting from reduced polar burden that facilitates passive membrane crossing. Surprisingly, the 14-mer population came second with a ratio of 3.3, suggesting that larger rings might offer unique advantages through conformational flexibility or chameleon-like behavior.
The Physicochemical Property Puzzle
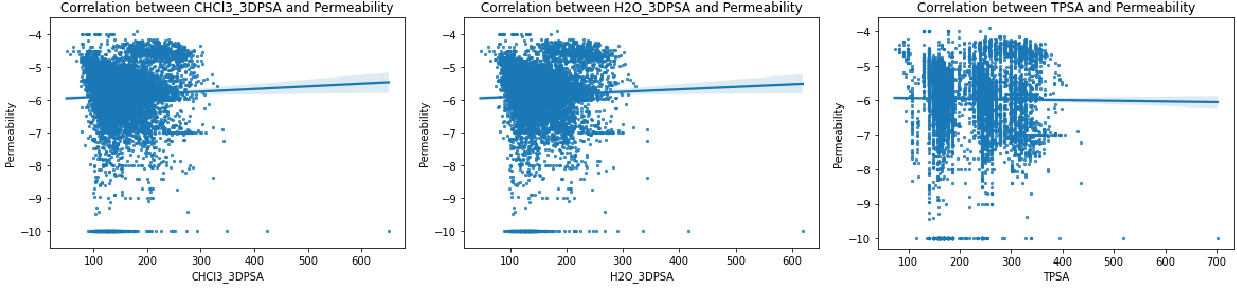
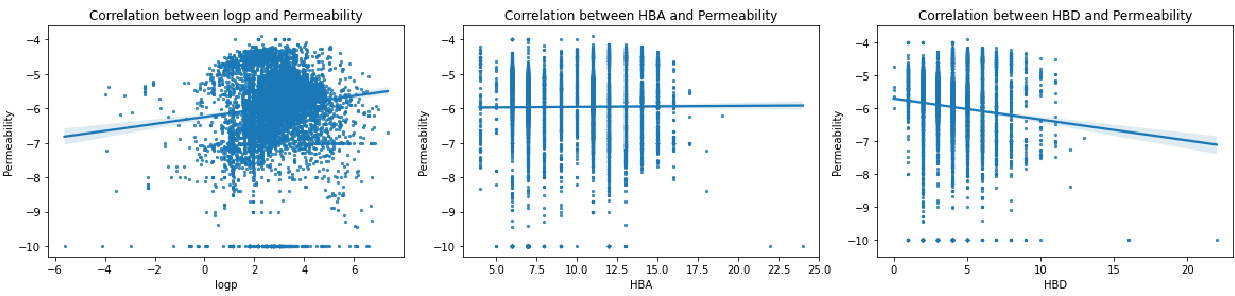
We systematically analyzed correlations between membrane permeability and key molecular descriptors: 3D polar surface area in chloroform and water (CHCl₃_3DPSA, H₂O_3DPSA), topological polar surface area (TPSA), lipophilicity (ClogP), hydrogen bond donors and acceptors (HBD, HBA), and polarity fraction (TPSA/MW).
Despite multiple literature reports positioning 3DPSA as a strong permeability predictor for other peptide systems [1-3], our analysis found no clear correlation. We suspect the reason is that database-generated 3D structures may miss the biologically relevant conformers that actually matter for membrane transit. This is particularly problematic for "chameleonic" cyclic peptides that dramatically reshape themselves when moving from aqueous to membrane environments.
ClogP, the traditional lipophilicity metric, also failed to show strong correlation with permeability, though most high-permeability peptides clustered within the 0-6 range. Similarly, hydrogen bonding capacity (HBD/HBA counts) proved surprisingly uninformative. Only the TPSA/MW ratio, essentially a polarity fraction, showed modest correlation with membrane crossing ability.
Summary: Navigating the Expanding Cyclic Peptide Data Universe
Today's journey through cyclic peptide databases uncovers a part of a rapidly expanding space. From CycPeptMPDB's focused permeability collection to NPMMPD's non-peptidic macrocycle universe to literature gems to comprehensive natural product catalogs, we now have unprecedented access to molecular diversity and experimental insights.
Yet this wealth of data also exposes fundamental gaps in our understanding. Traditional physicochemical descriptors, usually reliable guides in small-molecule drug discovery, stumble when met with the conformational complexity and chameleon-like behavior of cyclic peptides. The permeability puzzle remains partially unsolved, suggesting that we'll need new computational frameworks and experimental approaches to fully unlock the therapeutic potential of these molecular rings.
As the databases continue to grow and computational methods evolve, one truth emerges clearly: cyclic peptides occupy their own unique space with distinct rules that we're just beginning to decode. Having mapped the landscape of available data, we'll turn to a more strategic question in our next article: where should cyclic peptides make their mark?
Acknowledgments
Thank you to Yu Zhou (zhouy@dp.tech), Dongdong Wang (wangdd@dp.tech), and Jin(Tina) Yu (yujin@dp.tech) for their valuable contributions to the content for this article series.
References
[1] Rossi Sebastiano M, Doak BC, et al. Impact of Dynamically Exposed Polarity on Permeability and Solubility of Chameleonic Drugs Beyond the Rule of 5. J Med Chem. 2018 May 10;61(9):4189-4202.
[2] Poongavanam, V., Wieske, L.H.E., Peintner, S. et al. Molecular chameleons in drug discovery. Nat Rev Chem 8, 45–60 (2024).
[3] Le Roux A, et al. Marsault E. Structure-Permeability Relationship of Semipeptidic Macrocycles-Understanding and Optimizing Passive Permeability and Efflux Ratio. J Med Chem. 2020 Jul 9;63(13):6774-6783.