Inside the Ring Series 1: Introduction to Cyclic Peptides
Peptides: The Molecular Goldilocks
Peptides occupy a unique niche in the therapeutic landscape: molecularly situated between small-molecule drugs and large biologics. This intermediate size affords them a powerful combination of high target specificity and strong biological activity, often with reduced off-target effects. Small molecules remain foundational in pharmacotherapy due to their chemical diversity, oral bioavailability, and broad indications, particularly in oncology, cardiovascular, and immunological diseases. On the opposite end of the size spectrum, macromolecules like monoclonal antibodies provide highly specific mechanisms of action, typically deployed in complex immune-mediated conditions and certain cancers.
Peptides bridge these domains. Due to their intrinsic bioactivity and receptor-level precision, they offer an edge in metabolic disease, infectious disease, and immune-related disorders. The class encompasses naturally derived peptides (e.g., insulin), synthetically engineered analogs (e.g., liraglutide), and structurally modified variants (e.g., PEGylated interferons) designed for enhanced pharmacokinetic profiles.
Moreover, advances in solid-phase peptide synthesis, delivery systems, and stabilization strategies have significantly expanded the druggable landscape for peptides. What was once a challenging class — plagued by rapid degradation and poor bioavailability — has now become a focal point in modern drug discovery and translational medicine. As these innovations continue to evolve, peptide drugs are increasingly recognized not as niche agents, but as central pillars in the next generation of targeted therapeutics.
Cyclic Peptides: The Power of Circles
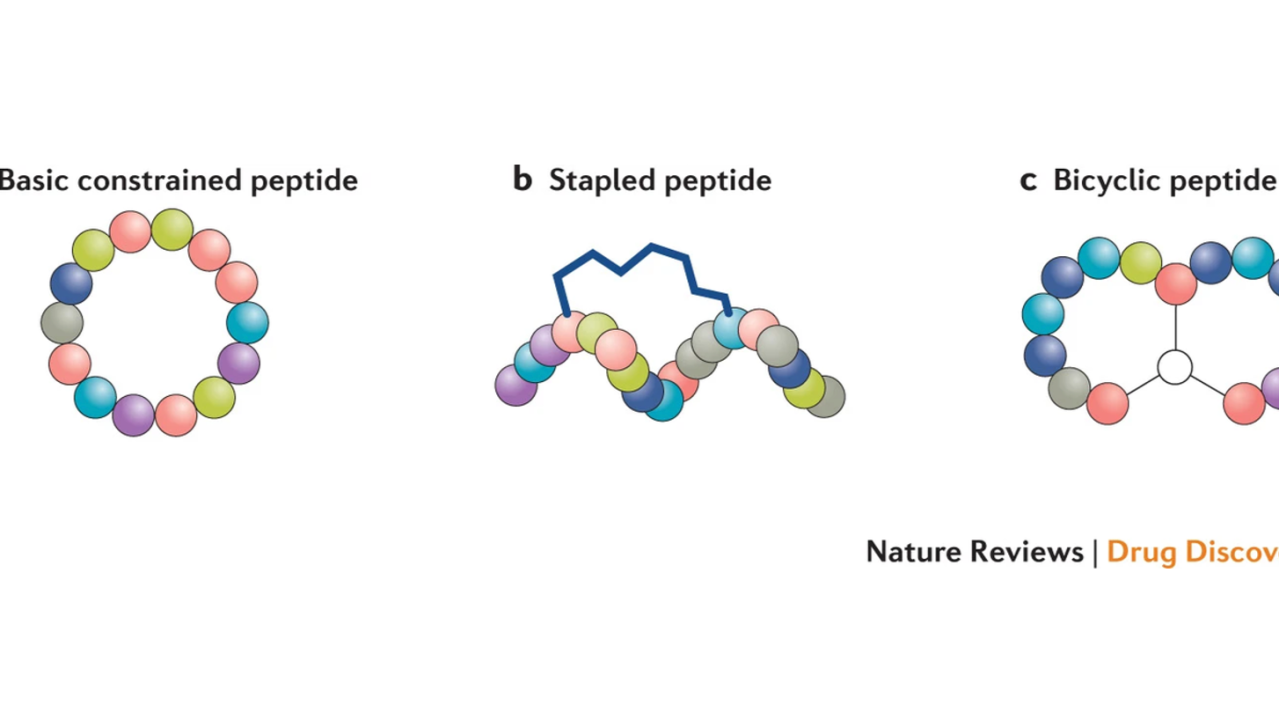
Cyclic peptides represent a distinct subclass of peptide therapeutics, characterized by covalently closed ring structures, either through head-to-tail cyclization or via side-chain linkages. This circular architecture imparts a number of pharmacologically desirable properties when compared with linear peptides. By restricting conformational flexibility, cyclization minimizes entropy loss upon target binding — a thermodynamic advantage that often enhances binding affinity. This rigidity also enables more precise spatial and electrostatic complementarity with molecular targets, improving selectivity and reducing off-target interactions. [1]
Critically, the structural constraints of cyclic peptides confer notable resistance to enzymatic degradation. Proteases, such as trypsin in the gastrointestinal tract or serine proteases circulating in the bloodstream, rely on specific substrate geometries to align with their catalytic triads and stabilize transition states. Cyclic peptides, with their limited backbone flexibility, often fail to conform to these requirements, and thus, evade proteolytic cleavage.
Additionally, the closure of the N- and C-termini effectively shields terminal polar groups from exopeptidase recognition, contributing to longer in vivo half-lives and higher metabolic stability. Cyclization also promotes intramolecular hydrogen bonding, which reduces solvent exposure of polar residues and masks parts of the polar surface. This in turn facilitates passive diffusion across hydrophobic barriers like cellular membranes, a major limitation for many linear peptides. While linear peptides can, in principle, adopt hydrogen-bonded structures, their higher conformational entropy renders such intramolecular interactions less thermodynamically favorable. In contrast, cyclic scaffolds are pre-organized to engage in these stabilizing interactions, further enhancing their pharmacokinetic and pharmacodynamic profiles.
Cyclic peptides can be classified according to multiple criteria, as follows [2–3]:
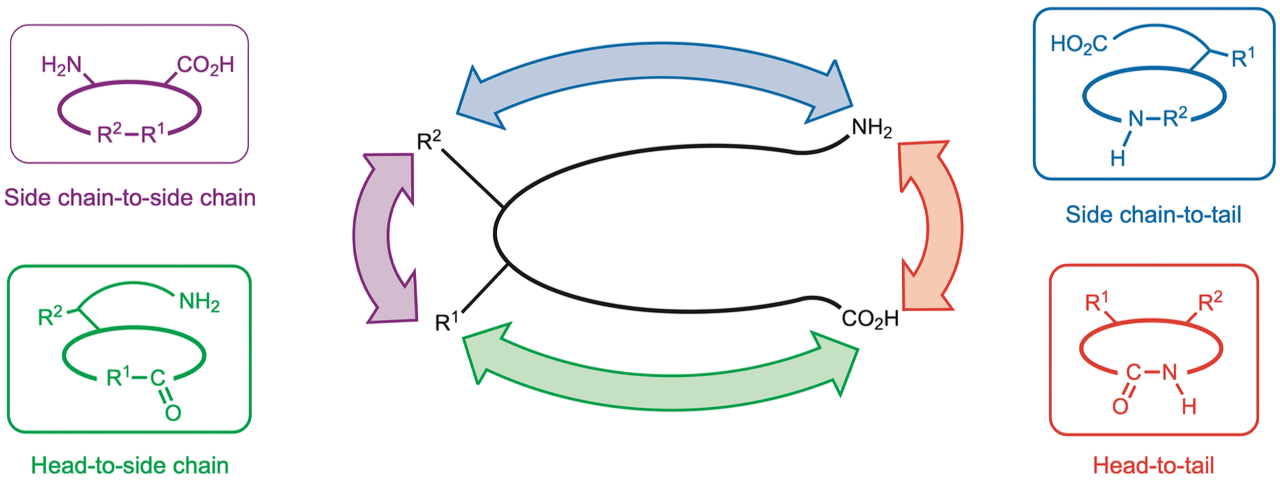
Size: Four or fewer amino acids are referred to as small cyclic peptides.
Mode of cyclization: Head-to-tail, head-to-side-chain, tail-to-side-chain, side-chain-to-side-chain, etc.
Number of backbone rings: Monocyclic, bicyclic, tricyclic peptides, etc.
Type of linkage:
- Homocyclic peptides (cyclized via amide bonds),
- Heterocyclic peptides (cyclized through non-amide linkages such as disulfide bonds, ester linkages as in lipopeptides, ether bonds, or thioether bonds).
The Good, the Bad and the Ugly
The Good
Cyclic peptides are seen as a therapeutic sweet spot that offers the best of both small molecules and large biologics. Structurally compact but functionally potent, they can engage protein–protein interfaces that are typically flat, shallow, and elusive to small molecules. This makes them especially attractive for "undruggable" targets. Compared with antibodies, cyclic peptides benefit from dramatically lower molecular weights that make navigating biological barriers much easier — penetrating solid tumors, reaching intracellular targets, and even crossing the blood–brain barrier.
The Bad
At the same time, their elegance comes with complexity. Although cyclization helps peptides slip past some of the permeability issues, the journey across a lipid membrane is a different story. Backbone amides and polar side chains continue to hinder passive diffusion, limiting cellular uptake without the help of specialized delivery systems or further chemical modification.
Then there’s the matter of synthesis. Cyclization doesn't always cooperate — it requires careful orchestration like amide bond formation or disulfide bridge establishment. Reaction efficiency can falter with longer sequences or bulky side chains, leading to low yields and scalability challenges. What looks like a tidy ring on paper can be deceptively hard to produce at scale.
The Ugly
Even once a cyclic peptide survives the challenge of design, synthesis, and delivery, it needs to be tested by in vivo stability. Despite resistance to enzymatic cleavage, many cyclic peptides are quickly swept out of circulation via renal filtration or hepatic metabolism. Their small size, while useful in tissue penetration, is prone to rapid clearance, which results in shorter half-lives and limited duration of action unless further optimized.
Key Moments in Cyclic Peptide Innovation
All in all, peptides have come a long way from lab curiosities to clinical mainstays. As of 2024, global regulatory agencies have approved 120 peptide-based therapeutics and diagnostic agents, and nearly half (46%) are cyclic peptides. This isn’t just a statistical footnote; it’s a reflection of how far the modality has evolved and the breadth of conditions these molecules can address.
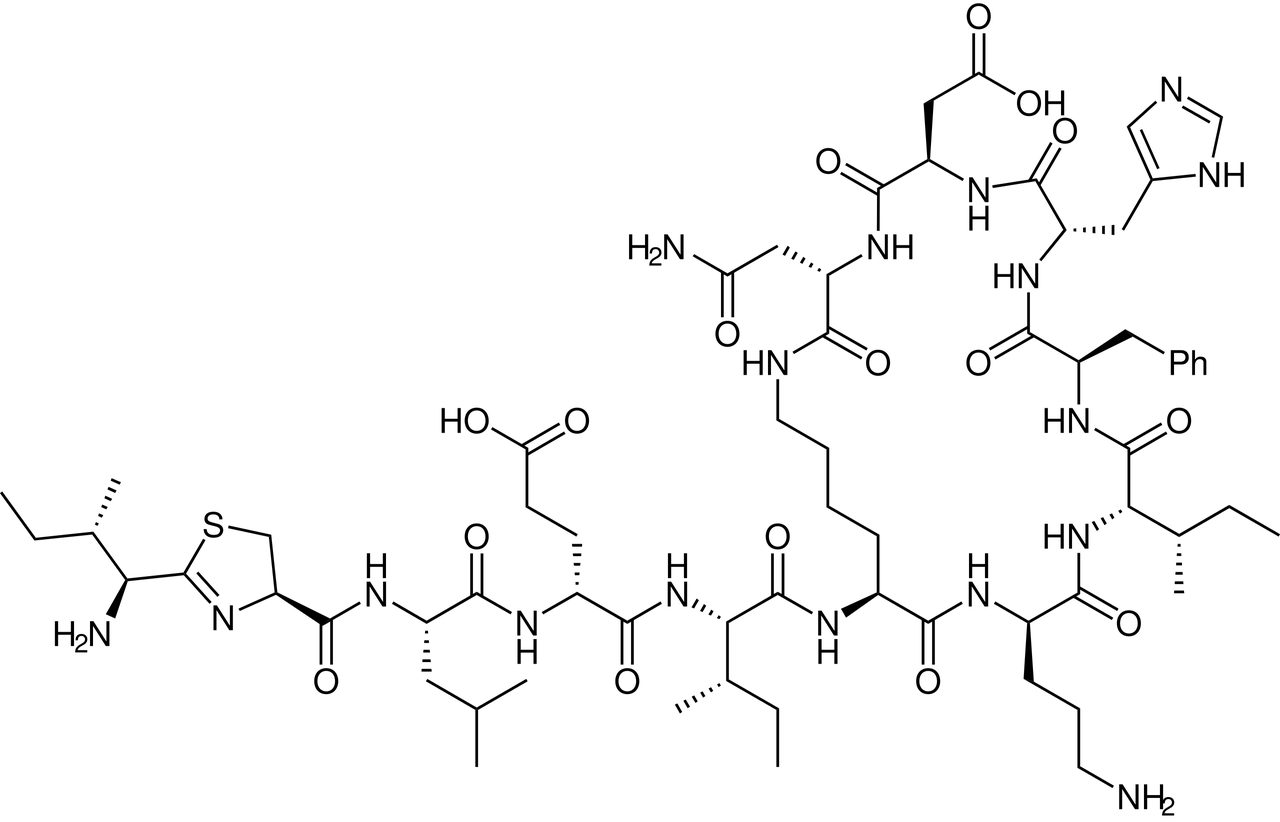
The broader therapeutic story began over a century ago with the clinical introduction of insulin in the 1920s. The cyclic peptide chapter officially began in 1942 with the discovery of gramicidin S. A 10–amino acid cyclic antibiotic produced by Bacillus brevis, it demonstrated broad-spectrum antimicrobial activity and became the first cyclic peptide developed as a drug.

Not long after, in 1948, the FDA approved bacitracin, making it the first cyclic peptide therapeutic to reach the market. Isolated from Bacillus subtilis and Bacillus licheniformis, bacitracin offered powerful antibacterial effects, particularly for skin infections, and became a household name in topical medicine.
Then came the breakthrough that changed the game: approved in 1983, cyclosporine A became the first orally available cyclic peptide. Derived from the fungus Tolypocladium inflatum, cyclosporine A acts as a calcineurin inhibitor, and is still widely used today to manage autoimmune diseases and prevent organ rejection. Its complex cyclic structure marked a turning point by proving that oral delivery, immunosuppression, and peptide-based drugs could coexist in a single molecule. These milestones paved the way for what cyclic peptides represent today: powerful, compact molecules with high selectivity, diverse mechanisms, and a growing list of clinical successes.
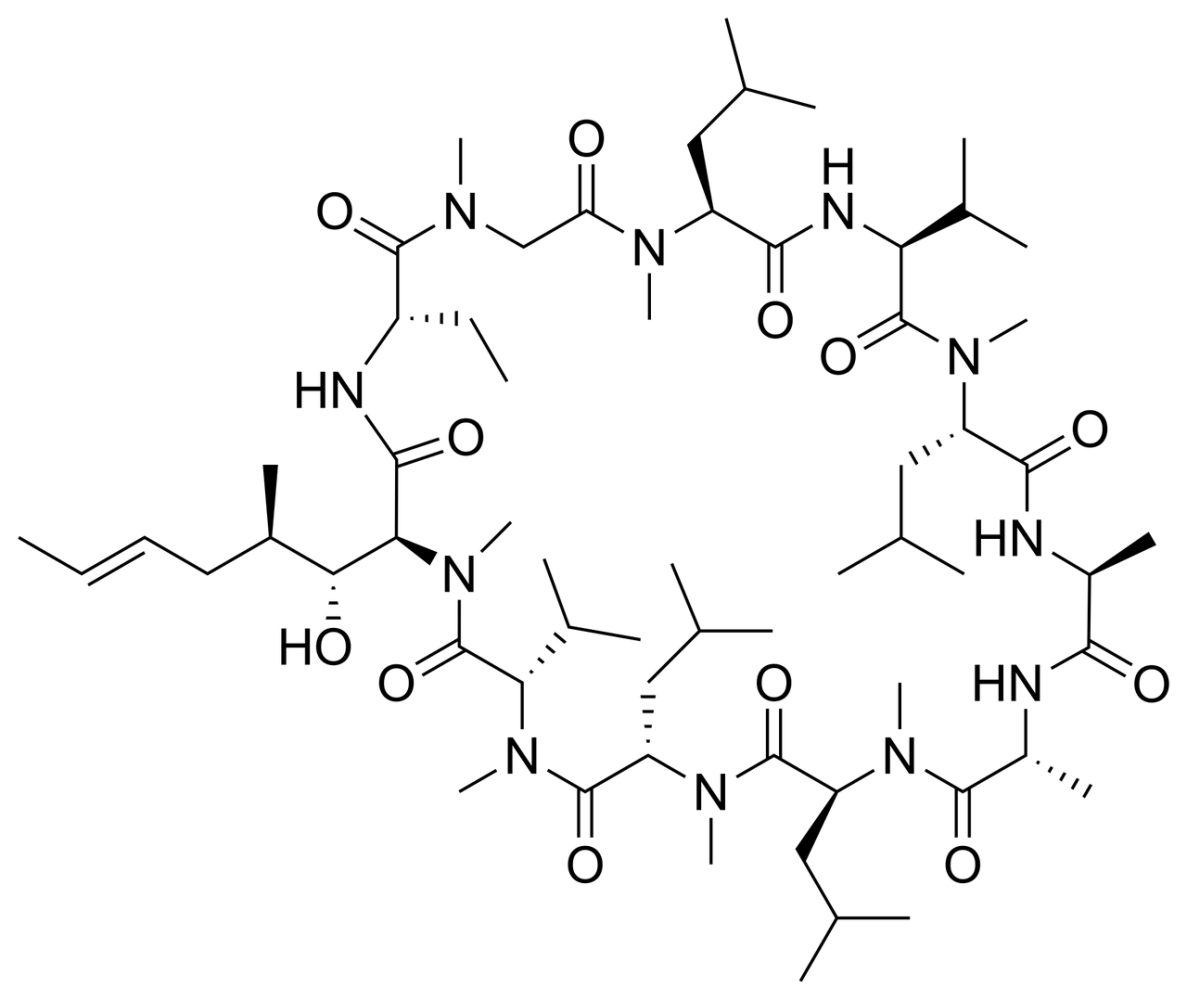
Over four decades have passed since cyclosporine A received FDA approval in 1983. Yet, despite substantial advances in cyclic peptide therapeutics, only a small fraction of these drugs are orally bioavailable today. The majority still depend on parenteral routes, a reality largely shaped by the inherent challenges of peptide chemistry and the harsh environment of the GI tract.
Peptides tend to be relatively large, highly polar, and structurally intricate, with features that limit their ability to passively cross the intestinal epithelium into systemic circulation. At the same time, a barrage of endogenous proteases in the gut rapidly degrades these molecules, further diminishing their oral bioavailability. Oral absorption of peptides is a complex interplay involving not only molecular size and stability but also factors like release kinetics, intestinal permeability, and first-pass metabolism in the liver. Overcoming these barriers requires more than just clever chemistry.

Encouragingly, recent years have seen significant progress. Innovations including strategic molecular modifications, co-administration of absorption enhancers, and sophisticated delivery vehicles are beginning to unlock the oral potential of peptide drugs. As these technologies continue to evolve, the goal of widely available, orally deliverable cyclic peptide therapeutics feels closer than ever — poised for the next breakthrough.

In our next article, we’ll dive into a brief overview of screening strategies, explore the Cyclic Peptide Database, and examine how permeability relates to key physicochemical properties.
Acknowledgments
Thank you to Zedong Wang (wangzd@dp.tech), Dongdong Wang (wangdd@dp.tech), and Jin (Tina) Yu (yujin@dp.tech) for their valuable contributions to content this series.
References
[1] Xinjian Ji, et al. Angewandte Chemie International Edition. 2024, 63, e202308251
[2] White, C et al. Nature chemistry, 2011, 3(7): 509-524
[3] https://www.zgbk.com/ecph/words?SiteID=1&ID=185288&Type=bkzyb&SubID=105294
[4] Lia Costa, et al. Pharmaceuticals, 2023, 16, 996
[5] https://peptherdia.herokuapp.com/
[6] Morrison, Chris. Nature reviews Drug discovery 17.8 (2018): 531-533.