Inside the Ring Series 1.5: From Lab Bench to Pill Bottle

This article of our Inside the Ring series spotlights the oral cyclic peptides already on the market, including recent entrants that have seized attention across our field.
Drugs Are More Than the Science
The impact of drugs reach beyond curing diseases: they reflect business realities, humanitarian imperatives, and societal responsibilities that extend far past their scientific foundations. The oral cyclic peptide market tells an especially compelling story of persistence against physiological odds. Today, we're focusing exclusively on oral formulations.
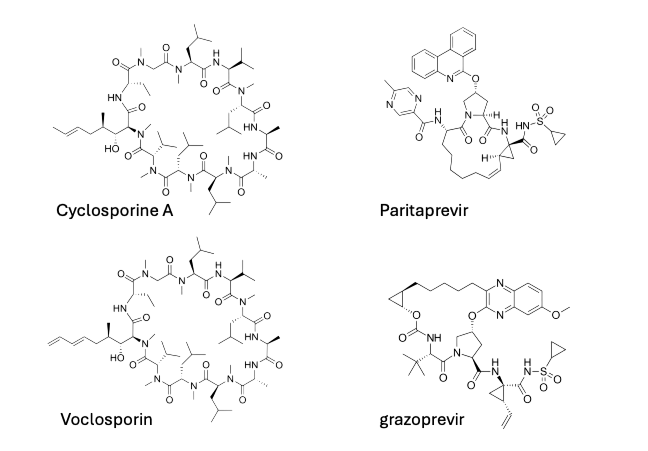
Marketed Oral Cyclic Peptides [1]
The evolution of orally administered peptide therapeutics is established on overcoming constraints in physiological design. Our GI tract, ruthlessly optimized for protein degradation over millions of years, poses a trinity of barriers: denaturing acidic pH, cleaving proteolytic enzymes, and blocking epithelial barriers. Yet, a select group of cyclic peptides has broken through these barriers by leveraging a sophisticated combination of structural optimization, formulation engineering, and deep understanding of pharmacology. The question isn't just how they succeeded, but what their success reveals about the future of peptide therapeutics in an increasingly competitive landscape.

High-Bioavailability Benchmarks
Within currently marketed oral cyclic peptides, several molecules show clinically meaningful systemic exposure. Cyclosporine A (Ciclosporin), a naturally occurring undecapeptide (Mw 1,202.64 Da), achieves ~30% bioavailability via conformational rigidity and high lipophilicity, enabling passive transcellular diffusion. Paritaprevir (Mw 765.89 Da) uses olefinic cyclization to lock its geometry and reach ~50% oral bioavailability, while grazoprevir (Mw 766.91 Da) uses a side-chain-to-tail cyclization to reach 27%; both target the hepatitis C virus NS3/4A protease. Finally, Voclosporin, a cyclosporin analogue (Mw 1,214.65 Da), incorporates a single amino acid modification that increases binding affinity 3–4-fold and nearly doubles oral bioavailability (~50%) compared to cyclosporine. These examples demonstrate how targeted chemical modifications can work with molecular topology to enhance absorption.
When Bioavailability Isn’t the Primary Success Indicator
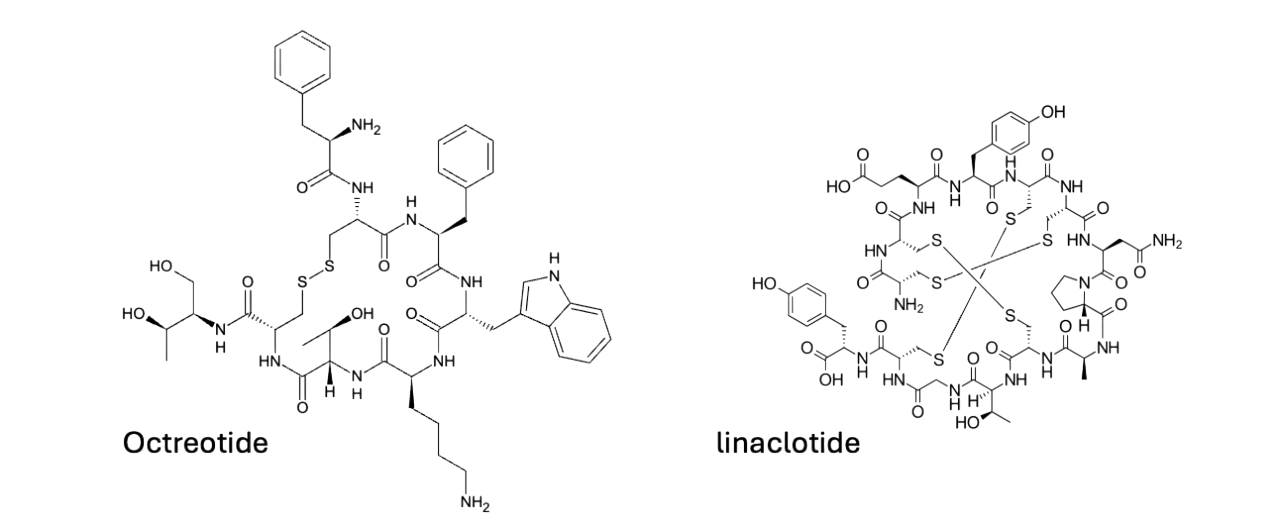
Most others fall far short of these numbers with <1% systemic exposure. Octreotide, a somatostatin analogue indicated for acromegaly, transitioned from parenteral (Novartis, 1988) to oral dosage form (Chiasma, 2020) by combining enteric protection with sodium caprate, which is a transient permeation enhancer that reversibly disrupts paracellular tight junctions in the small intestine. While still low, absorption is clinically sufficient for its pharmacodynamic target. Linaclotide, a 14-amino acid guanylate cyclase-C agonist for IBS-C and CIC, operates exclusively in the intestinal lumen. The negligibly low systemic exposure here is intentional, as local receptor activation increases chloride and bicarbonate secretion, and enhances motility without systemic effects.
These cases illustrate a spectrum of strategies:
- Structure-driven permeability (rigid macrocycles, lipophilicity tuning)
- Formulation-enabled uptake (enteric coatings, permeation enhancers)
- Pharmacology-aligned delivery (local vs systemic action)
A significant part of innovation in oral cyclic peptides will rely on aligning physicochemical properties with therapeutic objectives, rather than pursuing bioavailability as an absolute metric. Success will come from tailoring delivery strategies to the molecule’s mechanism of action, whether that means maximizing systemic exposure or exploiting local pharmacology to bypass absorption constraints.

Pipeline Contenders: Advancing the Oral Cyclic Peptide Frontier
A new generation of oral cyclic peptides is currently advancing through clinical development, each navigating the balance among molecular permeability, formulation strategy, and clinical ambition.
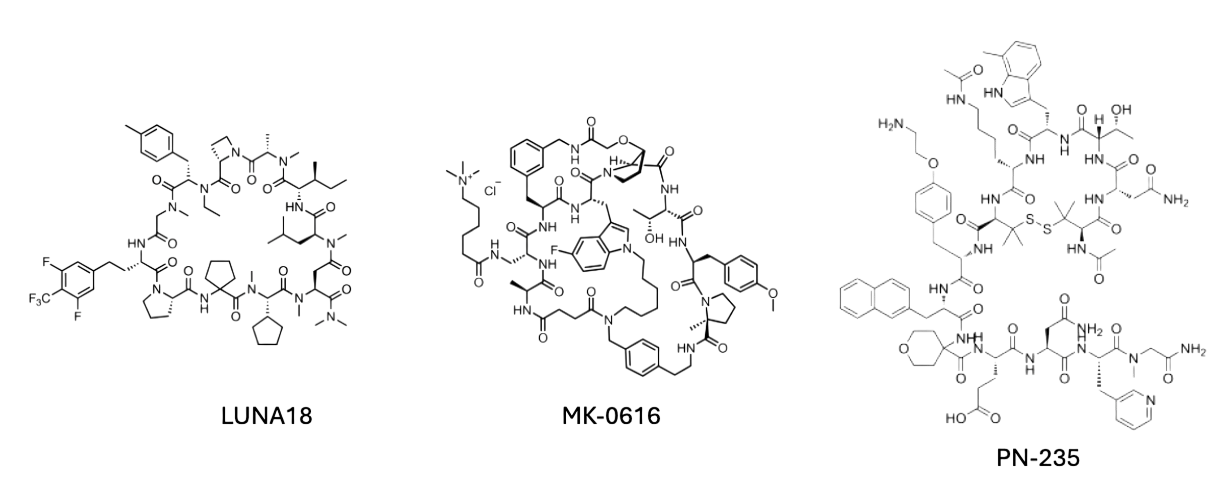
For example, Chugai’s LUNA18, a macrocyclic inhibitor targeting KRAS G12D, entered Phase I trials in Japan and the U.S. with early data showing favorable transcellular permeability and cross-species oral bioavailability ranging from 21–47% in mice, rats, dogs, and monkeys.[2] But on 24 July 2025, the program was suspended — a reminder that robust pharmacokinetics alone does not guarantee clinical or strategic viability.
Merck’s MK-0616, a macrocyclic PCSK9 antagonist in Phase III, faces the inverse problem: intrinsically low permeability that limits passive uptake. Its development hinges on pairing the molecule with a permeation enhancer, increasing estimated human bioavailability to ~2%, which is enough to meet LDL-cholesterol lowering goals in trials. [3,4]
Protagonist’s PN-235 (JNJ-2113) is an IL-23 receptor antagonist also in Phase III. Bioavailability in rats increased from 0.36% to 6.24% with an absorption-promoting excipient.[5,6] In July 2025, Johnson & Johnson announced submission of a marketing application for PN-235, potentially positioning it among the first of the next-generation oral macrocyclic immunomodulators.
These cases paint a clear picure: the road to successful oral peptide therapeutics is not a single paved path but a network of routes, some reliant on structural optimization to unlock passive permeability, others on formulation engineering to coax molecules across the gut wall, and still others on embracing local pharmacology where systemic exposure is unnecessary. The most successful strategies are those that match molecular design with therapeutic intent, ensuring that each peptide is delivered with purpose.
A Developing Story Based on Both Science and Strategy
The promise of oral cyclic peptides is not only reflected in clinical trials, but also in strategic business decisions shaping the space. In July 2025, Argenx struck a $1.5 billion deal with Unnatural Products to harness macrocyclic peptides against targets previously deemed “undruggable,” a move that further recognizes these molecules as versatile therapeutic platforms.[7] Earlier in April 2025, Merck & Co. signed a $493 million licensing agreement with Cyprumed, gaining access to its oral peptide delivery technology.[8] Further back in December 2024, PeptiDream demonstrated that oral macrocyclic myostatin inhibitors preserved muscle mass in mice when combined with semaglutide, signaling potential for combination therapy.[9]
Success in oral cyclic peptides is built from multiple puzzle pieces including chemical design, pharmacokinetics, strategic positioning, partnerships, and a firm grasp on market timing. Just as with molecules themselves, the path from concept to patients requires both scientific precision and entrepreneurial agility.
New to the series? Start with Inside the Ring Series 1: Introduction to Cyclic Peptides. In the next article, we'll dissect cyclic peptide databases to uncover the hidden relationships between molecular properties and membrane permeability.
Acknowledgments
Thank you to Zedong Wang (wangzd@dp.tech), Dongdong Wang (wangdd@dp.tech), and Jin(Tina) Yu (yujin@dp.tech) for their valuable contributions to the content in this series.
References
[1] https://peptherdia.herokuapp.com/
[2] Mikimasa Tanada, et al. Journal of the American Chemical Society. 2023, 145, 16610–16620
[3] Candice Alleyne, et al. Journal of Medicinal Chemistry. 2020, 63, 13796-13824
[4] Thomas J.Tucker et al. Journal of Medicinal Chemistry. 2021, 64, 16770-16800
[5] WO2021146441
[6] WO2022109328