From Antibodies to Peptides: How AOC and POC are Reshaping the Landscape of Nucleic Acid Drug Delivery
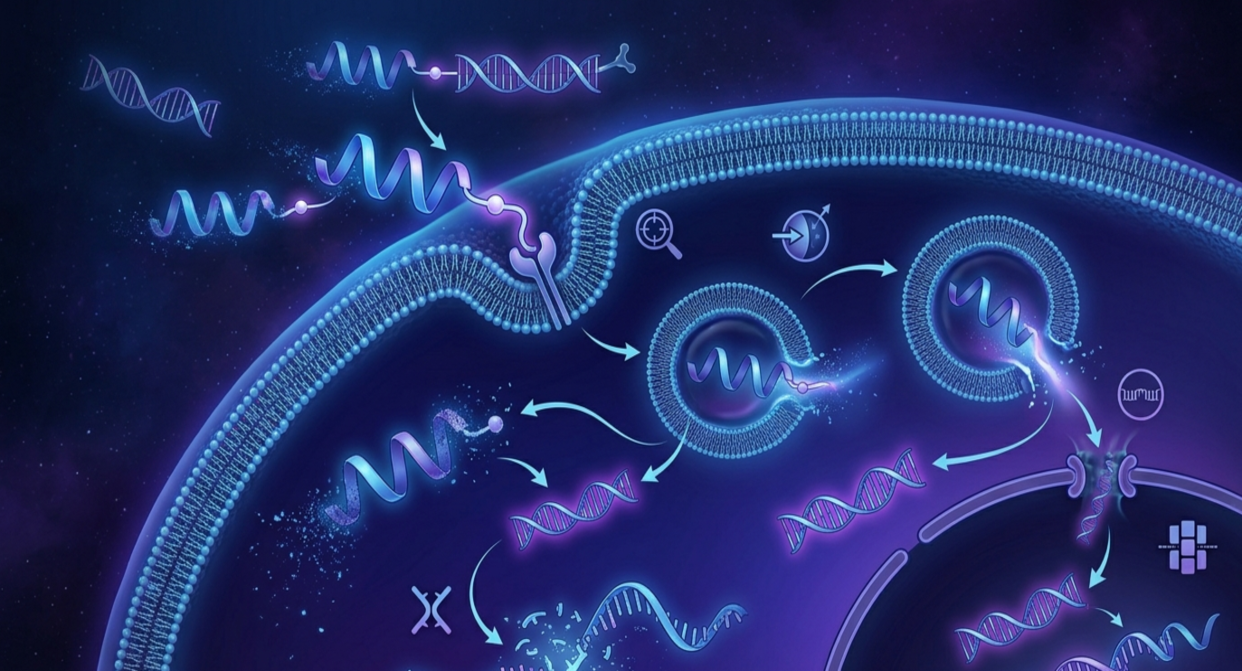
Background
1. The decisive battleground for nucleic acid drugs is delivery.
Although more than 20 nucleic acid therapeutics have been approved, most remain largely confined to the liver. Whoever can break through delivery barriers to muscle, heart, and the CNS will define the next frontier of the field.
2. CNS delivery: AOC leads in the clinic, POC aims for the future.
AOC (antibody–oligonucleotide conjugates)has more mature validation in crossing the blood-brain barrier (BBB). On the other hand, peptides, due to their extremely small size, theoretically offer more uniform diffusion within brain parenchyma, and may have the edge in expanding to new targets like LRP1.
3. Clinical divergence: AOC excels in potency, POC in penetration.
AOC demonstrates strong efficacy via precise receptor-mediated targeting (Avidity achieves ~40% exon skipping), while POC (peptide–oligonucleotide conjugates) shows a wider safety window and higher ceiling in dense muscle tissues (PepGen achieved 53.7% splicing correction at high doses).
4. The future is defined by the carrier and the target.
Choosing the right novel target (e.g., αvβ6) can achieve ~50% splicing correction at a very low dose (1.02 mg/kg), far exceeding TfR1-based approaches at the same dose. Thus, the next frontier will be based on scenario–target matching.
1. Conjugation Goes Beyond Linking
Conjugation in drug development is no longer just a chemistry problem, but is becoming a systems engineering challenge. The reason is simple: many conjugated drugs are not inactive; rather, they fail to reach the right tissue, the right cell, or even the correct subcellular location in a stable and precise way. This is particularly crucial for nucleic acid therapeutics. As of 2025, over 20 oligonucleotide drugs (including ASOs, siRNA, and aptamers) have been approved, validating nucleic acids. The next phase will hinge on not only sequence design, but also the delivery systems to transport these molecules beyond the liver into other tissues.
Peptides are re-emerging as key players in this delivery race. Their molecular weight sits in the goldilocks zone: smaller than antibodies to enable better tissue diffusion, yet more designable than small molecules to enable incorporation of targeting, membrane penetration, endosomal escape, and even subcellular localization functionalities. Peptides also align well with chemical synthesis logic, which allows for rapid iteration and scalable manufacturing. Beyond POC, peptide conjugation or fusion with small-molecule toxins, radionuclides, and proteins is expanding therapeutic modalities and continuously improving payload delivery, protein stability, solubility, delivery efficiency, and safety windows.
Oligonucleotide drugs come with inherent challenges including charge, poor membrane permeability, insufficient endosomal escape, and limited tissue distribution. Currently, AOC is more clinically mature, with strong data from late-stage pipeline leaders like Avidity and Dyne. Notably, Avidity was acquired by Novartis for $12 billion, signaling strong validation from capital markets and big pharma.
However, AOC’s maturity also points to limitations. Its advantages in high specificity, long half-life, and established biologics engineering are offset by drawbacks from the antibody itself: structural complexity, stringent CMC requirements, and limited tissue penetration due to size. To address this problem, researchers are leveraging smaller fragments like Fab and scFv.
Meanwhile, POC represents an entirely different delivery paradigm. Peptides can integrate targeting, membrane penetration, endosomal escape, and local enrichment into a shorter, more flexible carrier. Studies consistently show that peptides offer smaller size, easier manufacturing, enhanced tissue penetration, and improved cellular uptake, which are critical advantages for indications limited by tissue and cellular entry.
2. Defining the Optimal Battlefield for AOC vs. POC Mechanistically
2.1. BBB Crossing Validates AOC Effectiveness
Traditional antibodies enter the brain via the choroid plexus → CSF → perivascular spaces (PVS), then diffuse into parenchyma. This route leads to accumulation near vascular boundaries and interaction with vascular Aβ deposits, increasing the risk of ARIA (amyloid-related imaging abnormalities). On the other hand, AOC significantly reduces the risk of ARIA through TfR1-based “brain shuttle” strategies, which fundamentally alter this route by enabling receptor-mediated transcytosis across brain capillary endothelium. This mechanism increases parenchymal exposure while reducing vascular exposure and ARIA risk. Roche’s trontinemab is one example of this approach.
Meanwhile, POC platforms (e.g., Ionis/Bicycle) have shown BBB-crossing potential, but remain in early-stage development without landmark clinical pipelines.
The burning question: can POC replicate AOC’s success by targeting TfR1?
Intuitively, this seems plausible. In reality, the challenges are manifold. The critical factor is ensuring subsequent transcytosis, a highly dynamic process that imposes extremely stringent requirements on molecular design. The binding affinity must fall within a very narrow window: if too strong, the molecule risks degradation following endocytosis; if too weak, it fails to establish stable binding.
Simultaneously, it's critical to minimize valency to prevent receptor clustering, which would otherwise shunt the molecule toward the lysosomal degradation pathway. The challenge is to ensure transport of the molecule across the endothelial barrier (transcytosis), rather than being prematurely degraded by the endothelial cells. Thus, AOC’s advantage in brain delivery is not superior targeting, but rather, the ability to be engineered as a reliable transcytosis vehicle with strong safety control. However, this is not the endgame. Antibodies (~150 kDa) face diffusion limitations in dense brain tissue, while peptides (<5 kDa), after overcoming transcytosis barriers or leveraging new targets (e.g., LRP1 via Angiopep-2), can achieve far superior parenchymal distribution, and potentially enable POC to overtake AOC in later stages.
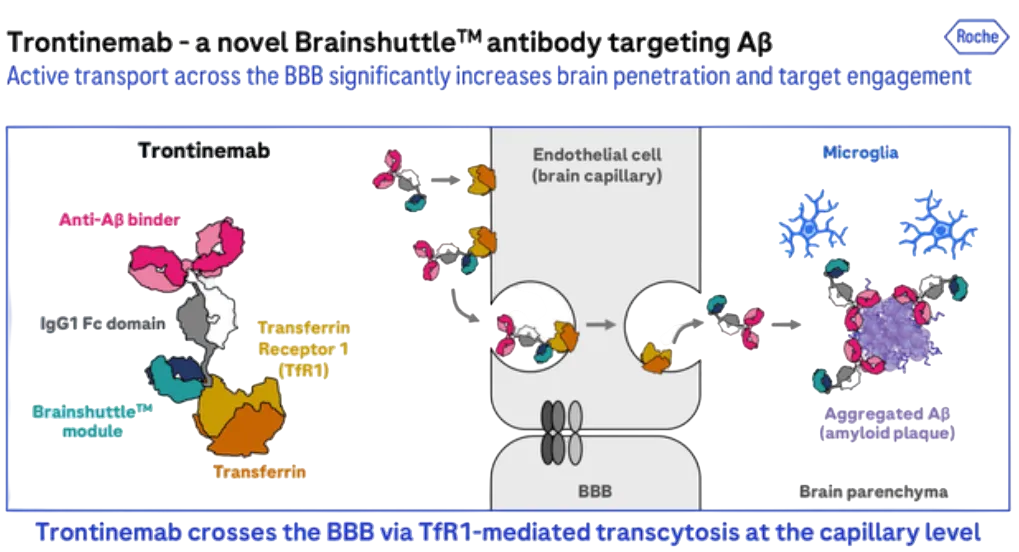
2.2. POC Shines in Muscle and Tumor Delivery
Diseases affecting muscle tissue differ fundamentally from those involving the brain. The core challenge lies in drug distribution and the extent of tissue coverage. For example, take rhabdomyolysis: a persistent hurdle has long been reaching a sufficient number of muscle fibers. Early gene therapies based on AAV (adeno-associated virus) have demonstrated this point: if only a subset of cells is targeted, the disease remains highly prone to recurrence even if initial efficacy is observed. Unless next-generation technologies can resolve this issue of comprehensive coverage, it will be difficult to achieve a true clinical breakthrough.
Compared to brain tissue, muscle tissue lacks a BBB and is devoid of spatial risk structures such as perivascular spaces (PVS). Consequently, drugs can typically reach the muscle interstitium without much trouble. But it's difficult for drugs to enter the muscle cells themselves. Muscle fibers are characteristically massive, multi-nucleated cells with highly stable cell membranes. Moreover, the cells are tightly packed together. The cumulative effect of these factors slows down the diffusion of large molecules like antibodies, resulting in a markedly heterogeneous distribution. This also explains why the therapeutic efficacy of AOCs in muscle tissue is somewhat limited. Although they can enter a subset of cells via receptor-mediated uptake, their large molecular size restricts their diffusion capacity within the tissue, making it difficult to achieve uniform coverage across the entire muscle tissue.
Here's where POC saves the day. Due to their smaller molecular size, peptides exhibit superior diffusion capabilities within tissues, enabling them to traverse the extracellular matrix (ECM) more readily and achieve a more uniform distribution throughout the muscle tissue. Furthermore, cell-penetrating peptides (CPPs) can enhance cellular uptake, thereby ensuring that a greater number of muscle cells are reached. Although this approach may lack absolute precision, it paradoxically becomes a distinct advantage in the treatment of indications requiring broad-spectrum tissue coverage.
The same reasoning applies to fibrotic diseases and solid tumors. Whether confronted with the dense ECM or complex micro environments, the diffusion of large molecules is invariably restricted, resulting in highly non-uniform drug delivery. In such scenarios, peptide-based delivery systems often stand a greater chance of achieving superior tissue penetration and coverage.
3. Comparing AOC vs. POC Based on Clinical Data
POC follows two distinct approaches. The first is "Receptor-Targeted POCs." The conceptual approach here is nearly identical to that of AOCs: both achieve organ-directed delivery by targeting tissue-specific receptors. Examples of this include Ionis and Bicycle’s TfR1 strategy, and Arrowhead’s αvβ6 targeting.
The other approach involves POCs that rely on Cell-Penetrating Peptides (CPPs). These candidates are supported by robust clinical data, allowing for a certain degree of comparison with current leaders in the AOC space, like Avidity, despite their fundamental differences in the underlying targeting strategies. Unlike receptor-mediated targeting, CPPs typically enter cells in a broad, non-specific manner. However, through specific medicinal chemistry modifications, they can be engineered to exhibit preferential distribution within muscle tissues.
Leveraging the inherent characteristics of muscle tissue, including its rich blood supply, high capillary density, and active endocytosis, CPPs can gain enter via multiple pathways, notably macropinocytosis and caveolae-mediated endocytosis. The subsequent challenges are effective endosomal escape and precise intracellular localization. Both Pepgen’s EDO (Enhanced Delivery Oligonucleotide) platform and Entrada’s EEV™ (Endosomal Escape Vehicle) platform are specifically focused on optimizing their technologies to address this key issue.
3.1. CPP-POC and AOC
AOC is currently more clinically advanced clinically (Avidity, Dyne in late-stage), while POC leaders (Entrada, PepGen) are in earlier stages.
DMD: Avidity vs. Entrada
Based on their specific indications and targets, we compare here the performance of Avidity’s del-zota against Entrada’s ENTR-601-44 in treating Duchenne muscular dystrophy (DMD): both therapies target Exon 44 skipping and employ a strategy of delivering PMOs. We also compare Pepgen’s PGN-EDODM1 against Dyne’s DYNE-101 for the treatment of myotonic dystrophy type 1 (DM1): while both target DMPK (Dystrophia Myotonica Protein Kinase), DYNE-101 delivers an ASO rather than a PMO (Phosphorodiamidate Morpholino Oligonucleotide).
Avidity
In October 2025, Novartis acquired Avidity for $12 billion, driven primarily by the appeal of three late-stage pipeline candidates targeting hereditary neuromuscular diseases. Avidity has developed a comprehensive AOC platform centered on TfR1, incorporating extensive optimizations in medicinal chemistry and CMC. For instance, by leveraging the electrically neutral properties of PMOs, the company has successfully pushed the Drug-to-Antibody Ratio (DAR) to exceptionally high levels, exceeding 9 in published literature, while maintaining safety. This achievement in itself demonstrates that AOCs possess an inherent advantage in terms of "drug-carrying capacity."

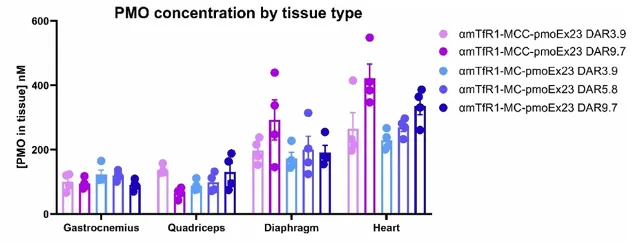
Furthermore, Avidity’s Del-zota (delpacibart zotadirsen) demonstrated clear biological activity in a Phase 1/2 study involving 44 patients with DMD: in both the 5 mg/kg and 10 mg/kg dose groups, post-treatment exon 44 skipping levels increased significantly, averaging approximately 37%–43% and reaching as high as approximately 67%. Concurrently, dystrophin protein levels also rose markedly, increasing by an average of 25%, with levels in some patients reaching over 50% of normal levels. Creatine kinase (CK), a biomarker of muscle damage, showed a sustained and significant decline of over 80%, approaching the normal range; this suggests that not only did molecular markers improve, but the underlying muscle pathology also underwent substantial amelioration.

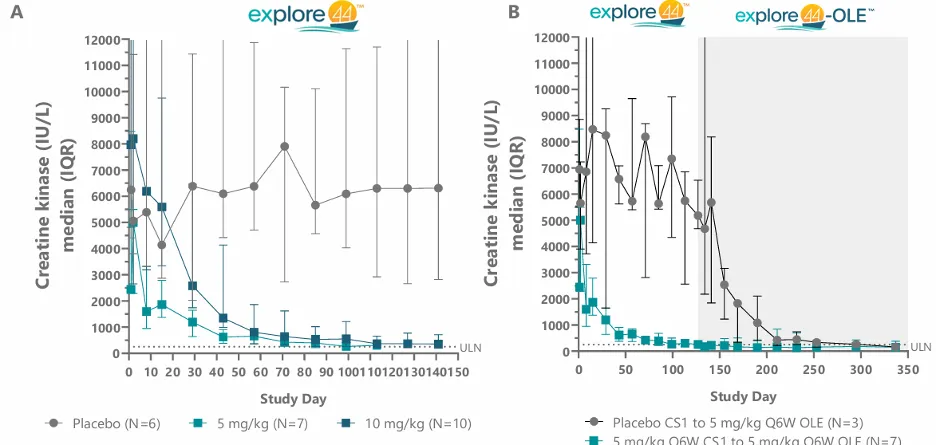
The majority of adverse events were mild to moderate in severity, while serious adverse events were infrequent, with no reported deaths or definitive dose-limiting toxicities. However, isolated serious adverse events (SAEs) were observed within the 5 mg/kg dose group, indicating that the safety window is a critical consideration as development progresses to higher dose levels.

Entrada
On the other hand, Entrada represents a distinct delivery paradigm on which the industry is placing serious bets. The company is pursuing an EEV (Endosomal Escape Vehicle) + PMO (Phosphorodiamidate Morpholino Oligomer) approach. The core philosophy here is to build its platform competitiveness around the "last mile" of intracellular delivery, specifically, endosomal escape, skeletal muscle distribution, and cardiac muscle delivery. To date, Entrada has advanced four clinical or late-stage preclinical programs targeting Duchenne Muscular Dystrophy (DMD) and is collaborating with Vertex on the DM1 program, VX-670. According to the company's public disclosures, its EEV-oligonucleotide system has demonstrated broad muscle distribution, significant cardiac muscle delivery, and the potential to nearly fully restore dystrophin production in skeletal muscle during preclinical studies.
Compared with Avidity’s Del-zota, Entrada’s clinical progress lags slightly; ENTR-601-44 is in the early stages of Phase I/II development at the time of this article, and patient efficacy data has not yet been disclosed. The only data released thus far consists of preclinical results, safety data from healthy volunteers, and biomarker data from healthy volunteers; efficacy data is not yet available. However, the information so far reveals several intriguing trends.
Although PMOs have stable and neutral physicochemical properties, the molecular weight of CPPs is significantly lower than that of antibodies. Consequently, the drug-loading capacity of POCs cannot be increased to the same extent as that of PMO-AOCs without the risk of disrupting the charge distribution of the CPP and conformation alteration. Therefore, the drug-loading capacity is set at one.
The highest dose level for ENTR-601-44 has reached 6 mg/kg, lower than that of Avidity, but with an exceptional safety profile. In the 6 mg/kg dose group, overall tolerability was good, with no observed drug-related adverse events. The most common adverse event (AE) was mild to moderate headache. There were no serious adverse events (SAEs), clinically significant abnormalities in laboratory parameters, ECGs, or vital signs. Further, renal toxicity biomarkers showed no abnormal signals even at the highest dose. Compared with Del-zota, which reported two SAEs in its 5 mg/kg dose group, ENTR-601-44 demonstrates a superior safety performance.
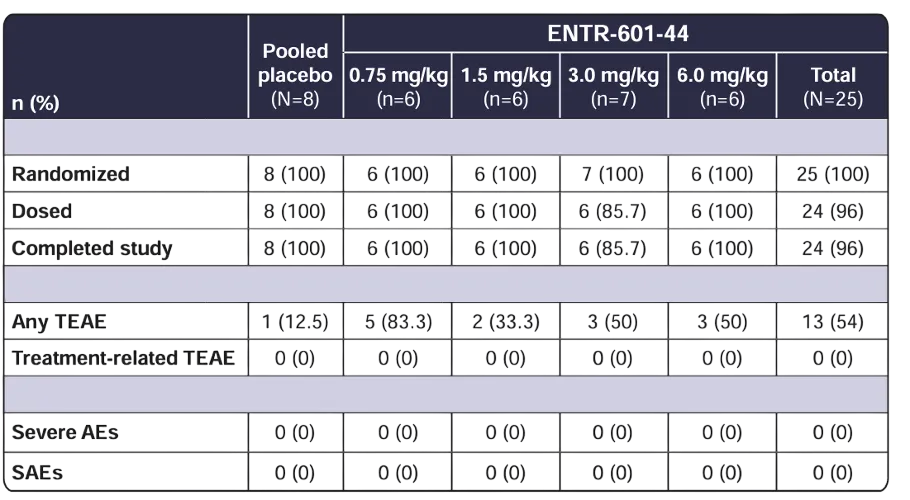
Given that the study subjects were healthy individuals, the efficacy findings are limited to biomarker data. In muscle biopsies performed 72 hours post-administration, quantifiable levels of the PMO-44 metabolite were detected in the *biceps brachii* of the 6 mg/kg dose group, averaging approximately 52.4 ng/g. More importantly, only the 6 mg/kg group demonstrated statistically significant DMD exon 44 skipping, averaging approximately 0.44%, compared to approximately 0.22% in the placebo group. While this value may not appear substantial in itself, its significance lies in the demonstration that, following a single intravenous administration in humans, a detectable on-target effect is indeed present within the muscle tissue.
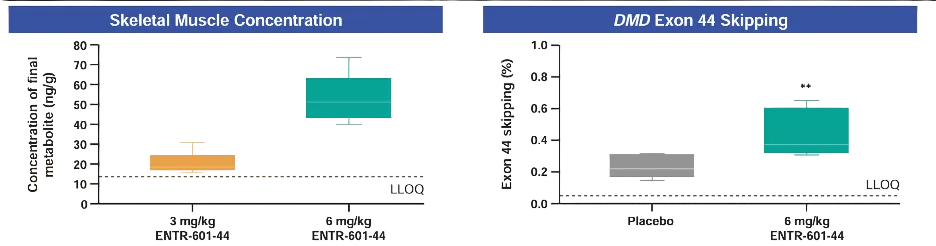
Also, recent animal studies demonstrate EEV-PMO’s ability to enter muscle satellite cells (Pax7+), exhibiting 100% co-localization with quiescent satellite cells within 48 hours. Even more remarkably, the accompanying figures indicate that PMO co-localization remains detectable within satellite cells and newly formed centrally nucleated fibers even after 12 weeks. This suggests that EEV does not merely target mature muscle fibers but may also access the muscle stem/progenitor cell pool—a factor of critical importance for long-term disease repair.

A clear conclusion emerges: at the current stage, AOC’s primary advantage is potency. Conversely, the strengths of CPP-POC are more evident in its broad distribution, safety profile, and potential for long-term tissue coverage. AOC focuses on solving the problem of how to precisely deliver the therapeutic agent, whereas CPP-POC addresses the possibility of delivering the agent to a wider range of cells.
- AOC (Avidity Del-zota):
- Exon skipping: ~37–43% (up to ~67%)
- Dystrophin increase: ~25% average, up to >50%
- CK reduction: >80%
- Safety: generally acceptable, some SAE at higher dose
- CPP-POC (Entrada ENTR-601-44):
- Early-stage, no patient efficacy yet
- Strong safety profile up to 6 mg/kg
- Detectable target engagement in human muscle
- Potential to reach satellite cells (important for long-term repair)
AOC = strong potency
CPP-POC = broader distribution, better safety, long-term potential
DM1: PepGen vs. Dyne
Another common indication for nucleic acid-based therapies is myotonic dystrophy type 1 (DM1). The underlying cause of DM1 is the presence of pathogenic CUG repeats (CUGexp) within the *DMPK* transcripts. These aberrant RNA molecules form hairpin structures that sequester MBNL1 (Muscleblind-like splicing regulator 1), a critical splicing factor, ultimately leading to widespread mis-splicing. For this specific indication, the design rationale for PMOs involves selectively binding to this pathogenic RNA sequence to disrupt hairpin formation and liberate MBNL1, thereby restoring normal splicing. Thus, the therapeutic target is the aberrant *DMPK* RNA itself, rather than the protein.
PepGen
PepGen represents a more "pure-play" approach to the POC/CPP pathway. At its core lies the Enhanced Delivery Oligonucleotide (EDO) platform, which uses optimized delivery peptides to enhance tissue penetration, cellular uptake, nuclear delivery, and endosomal escape. By the end of 2025, PepGen held approximately $148.5 million in cash on its balance sheet, enough to sustain operations through the second half of 2027, but unlike Entrada, has yet to secure a major partnership endorsement from a large pharmaceutical company.
PepGen’s PGN-EDODM1 has completed Phase 1, demonstrating good tolerability. The highest dose cohort tested at 15 mg/kg exceeded the maximum doses administered in currently marketed AOCs. The most common treatment-emergent adverse events (TEAEs) included nausea, nasopharyngitis, and headache, mostly mild to moderate in severity. No TEAEs led to treatment discontinuation or any fatalities. Moreover, no electrolyte abnormalities like hypomagnesemia were reported.
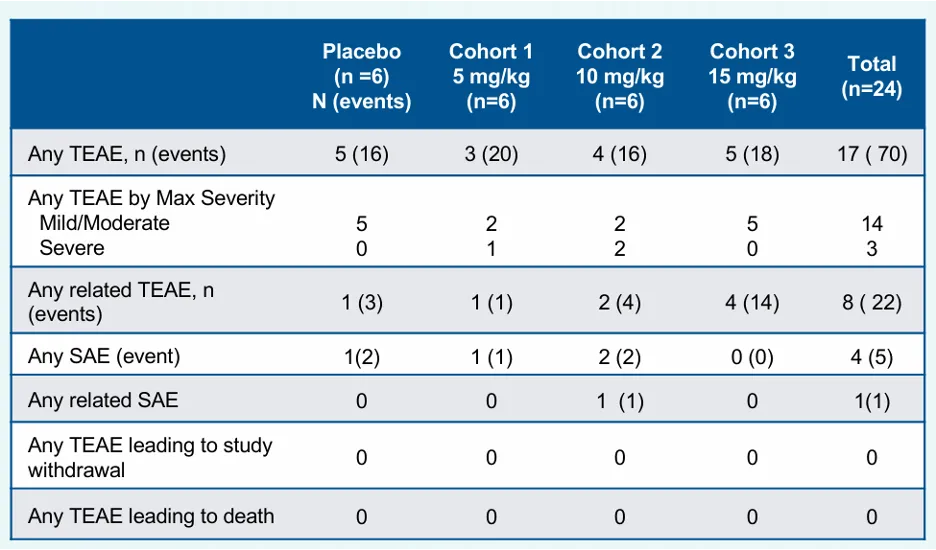
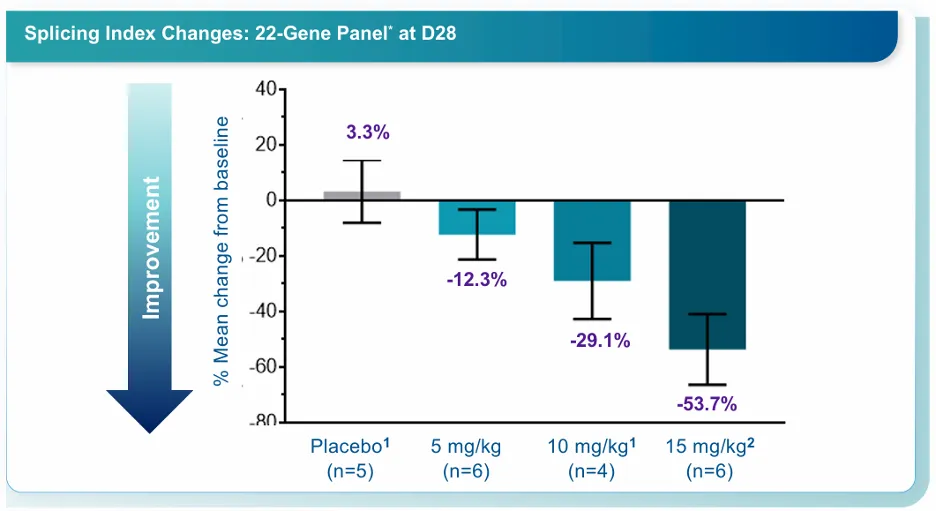
Efficacy was dose-dependent, with higher doses resulting in stronger splicing correction. Splicing improvement was observed in 87.5% of subjects across all dose levels, representing the highest average splicing correction reported to date for the DM1 indication. Given that patients in the high-dose group experienced transient declines in eGFR and increases in creatinine and albuminuria, albeit with a rapid return to normal levels, PepGen conservatively set the maximum dose for its Phase 2 clinical trial at 12.5 mg/kg.
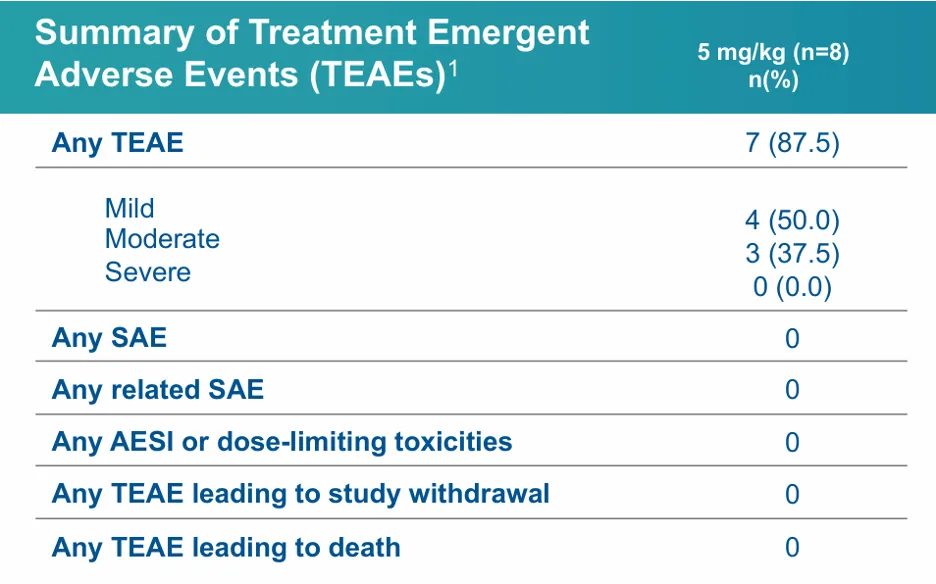
Results for the first dose cohort (5 mg/kg) of the PGN-EDO Phase 2 clinical trial were recently released. In this cohort, the treatment was generally well-tolerated. All adverse events (AEs) were mild or moderate, and there were no serious adverse events (SAEs), dose-limiting toxicities (DLTs), hypersensitivity reactions, or renal-related treatment-emergent adverse events (TEAEs). Most importantly, all 8 subjects completed the full course of 4 doses, and no cumulative toxicity was observed.
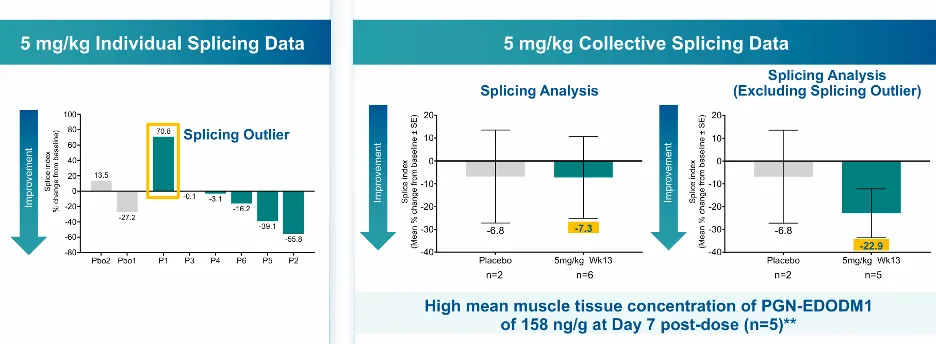
Regarding the functional assessment of finger relaxation speed, PepGen indicated a promising trend at the lowest dose of 5 mg/kg. The treatment group generally trended toward improvement compared with the placebo group throughout the follow-up period. This trend is more distinct when splicing outliers are excluded, and serve as a positive indicator of greater efficacy in subsequent, higher-dose cohorts.

Dyne Therapeutics
Unlike Avidity, which uses full-length antibodies (Fab + Fc), Dyne uses Fab-only antibodies. The company appears to be strategically focused on "productizing and platformizing" the AOC approach. Dyne has established around its FORCE platform a comprehensive delivery system comprising TfR1-binding Fabs conjugated to specific payloads. This system aims to maximize tissue penetration, optimize CMC (Chemistry, Manufacturing, and Controls) properties, and enhance platform scalability, while preserving TfR1-mediated muscle-targeting capabilities.
According to the Phase 2 clinical data for DYNE-101, the overall safety profile was acceptable, though a number of treatment-emergent adverse events (TEAEs) were observed. Across all cohorts, 100% of subjects experienced at least one TEAE. However, there were no serious treatment-related TEAEs, discontinuations due to adverse events, or fatalities. The most common TEAEs reported were nasopharyngitis (41%), procedure-related pain (34%), influenza (30%), infusion-related reactions (29%), headache (27%), and diarrhea (23%). Overall, even though the highest dose (6.8 mg/kg) was lower than those achieved by Pepgen or Avidity, multiple TEAEs were already evident, suggesting that its safety profile is slightly inferior to that of PGN-EDODM1.

Performance trends move in a favorable direction, with data indicating that at 12 months, the 6.8 mg/kg Q8W group outperformed the placebo group across multiple clinical endpoints. Objective functional measures, including the 10-meter walk/run test, QMT total score, middle-finger vHOT, 5-times sit-to-stand test, and 9-hole peg test, all demonstrated favorable changes.
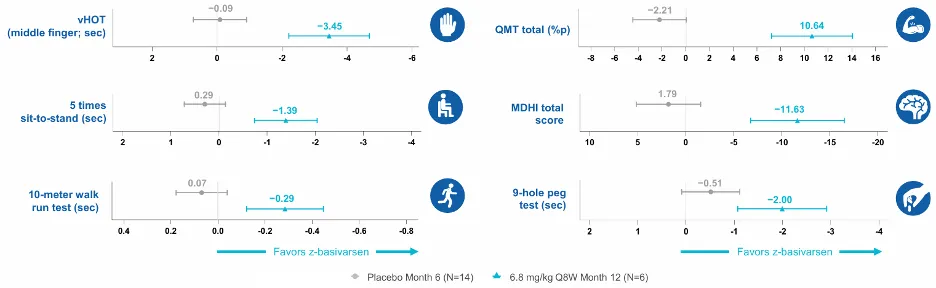
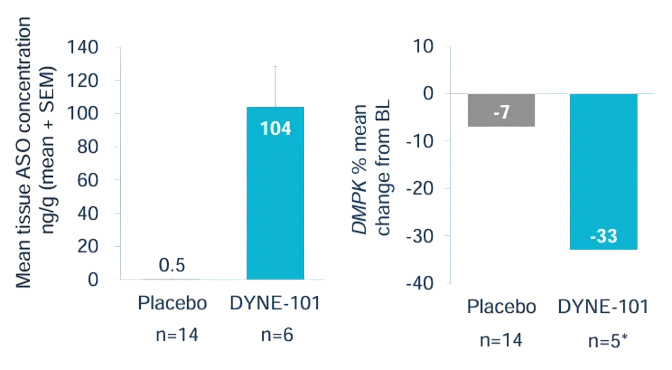
While notable efficacy was observed, the percentage improvement in the highest-dose group stood at only 33%, significantly lower than that of the highest-dose groups for Pepgen (53%) and Avidity (67%), indicating a lack of significant advantage in both safety and efficacy.
- PepGen (CPP-POC):
- Dose up to 15 mg/kg
- Splicing correction: up to 53.7% (highest reported)
- Strong dose-response
- Good safety, mild reversible renal signals
- Dyne (AOC-like Fab platform):
- Max dose: 6.8 mg/kg
- Splicing correction: ~33%
- Functional improvements observed
- More TEAEs, slightly weaker safety
3.2. Receptor-Targeted POC
The proof-of-concept (POC) collaboration between Ionis and Bicycle substitutes antibodies with a targeted peptide based on the TfR1 receptor. The ligand ultimately identified through their screening process is BCY17901, a bicyclic peptide (Bicycle) of approximately 2 kDa. When conjugated to an ASO or siRNA, this ligand significantly enhances muscle delivery and targeted mRNA knockdown in human TfR1 knock-in mice and non-human primates (NHPs).

Through a combination of phage display and structural biology, the researchers initially screened a library of Bicycle bicyclic peptides to identify candidates capable of binding to human TfR1. These candidates were then optimized through crystallographic analysis and alanine scanning. The identified binding site on TfR1 is a novel epitope — distinct from antibody-binding sites — that does not compete with transferrin for binding (the PDB ID for the crystal structure is 9GH7). Furthermore, the binding affinity was successfully optimized to the nanomolar to sub-micromolar range. Ultimately, BCY17901 was selected as the lead ligand, as it demonstrated both high specificity for human TfR1 and favorable properties regarding chemical synthesis and conjugation.
In an *in vivo* screening study conducted in human TfR1 knock-in mice, the research team synthesized conjugates of 12 distinct Bicycle–Dmpk ASO candidates and compared their efficacy in achieving Dmpk knockdown within the limb muscles and the heart. When compared at an equivalent dose of 3.5 mg/kg (based on ASO content), BCY17901 demonstrated significantly better inhibitory effects than the unconjugated ("naked") ASO.

Despite encouraging delivery efficiency and target knockdown effects in mouse and non-human primate models, receptor-targeted POCs remain in the preclinical stage; the available data mainly reflects the platform's delivery potential within animal systems. Given the significant interspecies variability in the delivery, cellular uptake, and sub-cellular distribution of nucleic acid therapeutics within muscle tissue, the predictive value of such animal data regarding human therapeutic efficacy is still limited.
In contrast, technological approaches like AOCs and CPP-POCs have already accumulated a substantial body of human clinical data, providing preliminary validation of their delivery efficiency, safety profiles, and the translatability of functional improvements. Based on current data, Bicycle's approach remains in the mechanistic validation phase and is not yet directly comparable to platforms already supported by established clinical evidence.
Ionis/Bicycle developed TfR1-targeting bicyclic peptide (BCY17901):
- ~2 kDa peptide ligand
- Strong binding to human TfR1 (non-overlapping epitope)
- Improved muscle delivery and mRNA knockdown in mice/NHPs
3.3. Target Selection Matters
Sarepta and Arrowhead also have a POC program targeting DM1, with their αvβ6 target demonstrating impressive progress. While αvβ6 has long been deemed a target for fibrosis, it has unexpectedly indicated outstanding potential in the context of muscle delivery. In the 1.02 mg/kg dosage group alone, the improvement rate reached 50%, far exceeding those of all currently available TfR1-based approaches.

At times, stepping away from the intense competition surrounding popular targets can yield unexpected and remarkable results. At the same time, given the novelty of αvβ6 as a delivery target and the lack of benchmark competitors in the market, direct head-to-head comparisons remains a challenge.
4. From Platform Competition to Scenario Matching
In terms of clinical maturity, AOC currently maintains a slight lead overall. Avidity and Dyne have already presented robust human data from late-stage or advanced clinical trials, indicating robust industry validation. In contrast, the CPP-POC approach lags slightly in the depth of its human validation but demonstrate distinct safety profiles, tissue distribution capabilities, and potential for sustained long-term coverage. This is especially important when the delivery target extends beyond mature muscle fibers to encompass more critical cellular populations like satellite cells and progenitor cell pools.
In the long run, AOCs and POCs will each establish its own dominance across different therapeutic scenarios:
- AOC: best for high specificity and receptor-mediated delivery (e.g., BBB crossing)
- POC: best for broad distribution and deep tissue penetration (e.g., muscle, tumors, brain parenchyma)
The ultimate potential of a delivery system often hinges on selecting the right entry point, rather than simply swapping out the carrier. From this angle, the historical mission of receptor-targeted POCs needs to extend beyond simple substitution for AOCs to leveraging their molecular advantages to explore entirely new gateways into tissues and identify novel equilibrium points for pharmacokinetics and pharmacodynamics (PK/PD).
The truly exciting part is not a zero-sum game of who replaces whom, but rather a paradigm shift in delivery technology — one that shifts from single-platform competition toward physics- and biology-driven, scenario-specific stratified evolution, where delivery technologies are matched precisely to biological constraints and therapeutic goals.
Acknowledgments
Thank you to our authors: Xinyu Zhong, Jiayue Wang, Merlin Wu, Dongdong Wang, and Yiwei Fu.